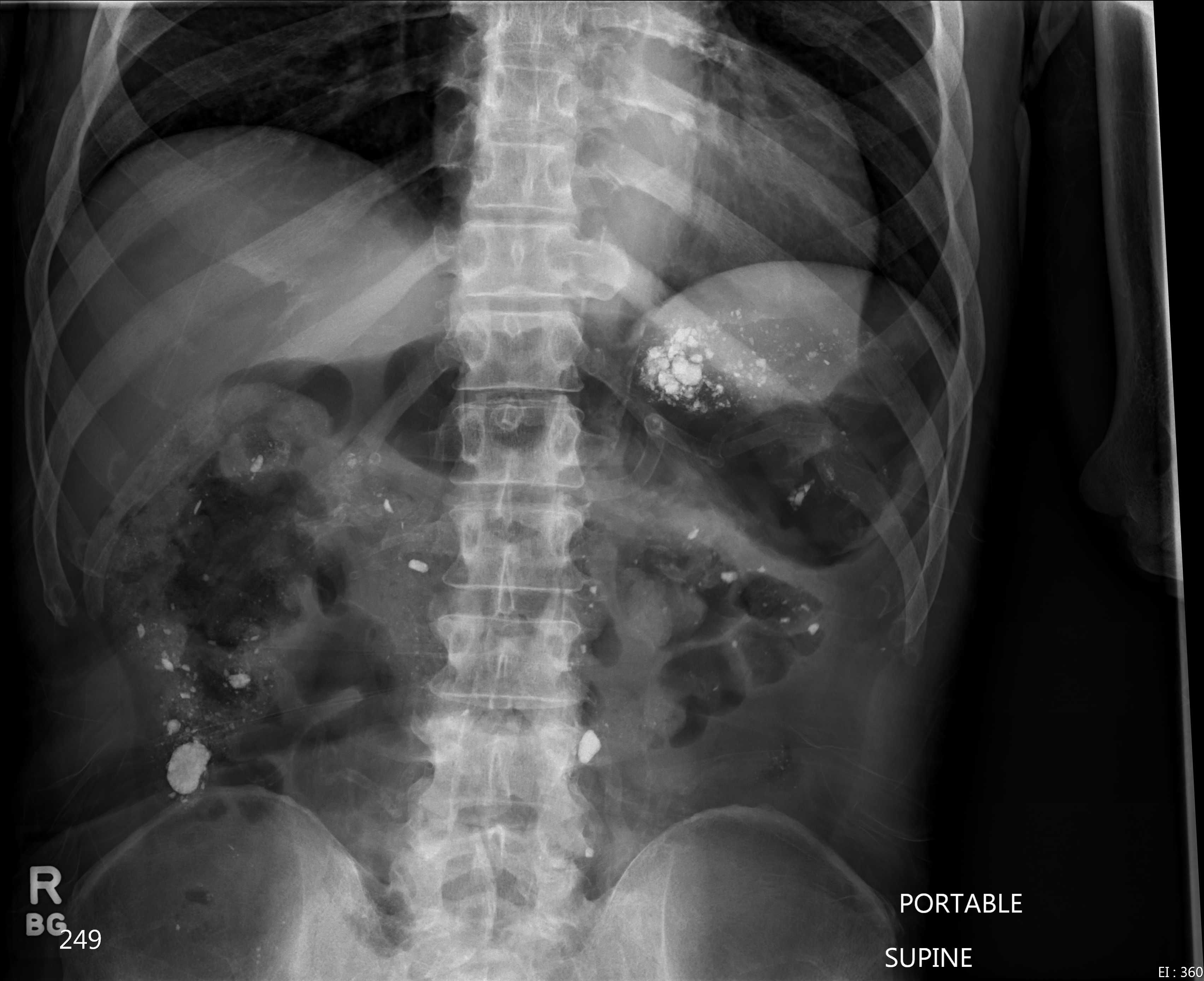
Case Presentation:
A 68 year old male with history of hemodialysis dependent chronic kidney disease, stroke and vascular dementia was transferred from a nursing home with increased lethargy and abdominal pain. In the emergency department he was found to be febrile to 101.9°F and abdominal exam revealed involuntary guarding with diffuse pain to deep palpation. Laboratory investigation revealed a neutrophil predominant leukocytosis of 22,300/µL. A KUB did not reveal evidence of mechanical bowel obstruction but noted scattered high-density material in the abdomen reported to represent residual barium. The patient was admitted to the inpatient medicine ward, blood cultures were collected and empiric antimicrobial therapy was initiated with vancomycin, levofloxacin and aztreonam. Medication reconciliation by the primary admitting providers identified the patient was on lanthanum carbonate (LC) therapy but an extensive search failed to illicit a recent history of any barium administration or ingestion.
Six blood culture bottles returned positive for gram positive cocci (GPC) in pairs and chains within 24 hours of admission. GPCs were identified as vancomycin sensitive Enterococcus faecalis. The patient’s clinical condition rapidly deteriorated resulting in death from enterococcal sepsis on hospital day 2.
Discussion:
Lanthanum carbonate (FDA approved in 2004) is a poorly absorbed non-calcium, non-aluminum phosphate binder commonly prescribed in patients with end stage renal disease. Available as a chewable tablet and oral powder, LC reduces serum phosphate concentrations by binding to dietary phosphate forming an insoluble compound, lanthanum phosphate, in the gut. Lanthanum is a trivalent cation rare earth metal (atomic number 57) with similar radiologic properties as another earth metal, barium (atomic number 56). In this case, the radiopaque lanthanum deposits on abdominal radiograph were incorrectly identified as residual barium contrast. These radiographic properties highlight the importance of medication reconciliation to prevent diagnostic misinterpretations. LC should be stopped if elective abdominal imaging studies are to be performed.
Lanthanum carbonate is generally well tolerated with the most commonly reported adverse effects being mild gastrointestinal (GI) intolerance. However, both the tablet and oral powder formulations have been associated with fecalith formation, ileus and colonic perforation, particularly in the presence of known diverticular disease. Histopathologic studies have also demonstrated lanthanum deposition in gastric and colonic mucosa. In this case, the presence of high grade enterococcal bacteremia implicates a biologically plausible source of bacterial translocation via a GI source.
Conclusions:
Hospitalists need to be aware that lanthanum may appear as radiopaque densities on abdominal imaging. While rare, serious adverse events could lead to potentially fatal GI complications.