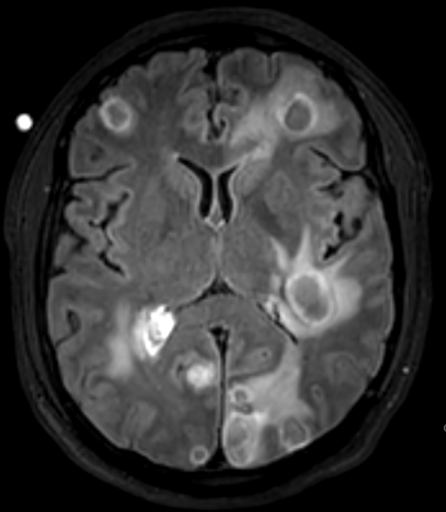
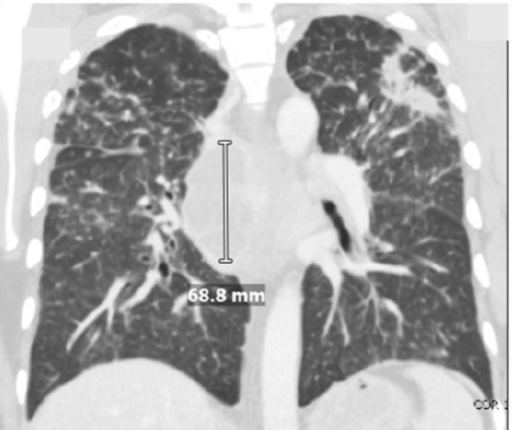
Case Presentation: A 41-year-old previously healthy man presented with three weeks of cough and one week of progressive headache, fever, confusion, and weakness. He is currently a construction worker who was born in Guatemala but has lived in Southern California since 2001. Initial vital signs were notable for a fever of 102.7 F, pulse 134, respirations 18, and BP 120/78. Initial labs showed WBC 20.9, Na 125, ESR 70, CRP 1.4, HIV negative, CD4 579, and CD8 145. Brain MRI revealed innumerable ring-enhancing lesions, and CT Chest showed multiple mediastinal lymph nodes with necrosis and bilateral upper lobe consolidations (Figures 1 and 2 respectively). ID and pulmonary were then consulted and recommended a bronchoscopy. Broad-range PCR on sputum cultures identified Nocardia cyriacigeorgica and Aspergillus. Given that preliminary testing with sputum culture was negative for tuberculosis and bronchoalveolar lavage was negative for acid-fast bacilli, neurosurgery was consulted, and the patient underwent a brain biopsy. Metagenomic next-generation sequencing of the cerebrospinal fluid studies and biopsy confirmed central nervous system (CNS) dissemination of Nocardia. Cultures from his expectorated sputum and brain biopsy grew Nocardia cyriacigeorgica. Therapy with empiric IV sulfamethoxazole-trimethoprim and imipenem was initiated. Imipenem was later switched to linezolid when susceptibilities became available. He then had a full immunodeficiency workup which came back positive for autoantibodies against anti-granulocyte-macrophage colony-stimulating factor (GM-CSF).
Discussion: Disseminated nocardiosis with involvement of the CNS is a rare yet severe infection that typically affects immunocompromised individuals. However, emerging evidence suggests that anti-cytokine autoimmunity, i.e. GM-CSF autoantibodies, may be a risk factor for opportunistic infections in patients without overt immunodeficiency or comorbidities [1-2]. These autoantibodies neutralize GM-CSF, a hematopoietic growth factor critical to the production and function of granulocytes and macrophages, leading to impaired innate immune response and phagocytosis, thus increasing susceptibility to nocardiosis and other opportunistic infections [3]. Intracerebral nocardiosis can cause severe disease in immunocompromised patients and accounts for up to 26 percent of nocardiosis cases [4]. Given disseminated nocardiosis, combination therapy is recommended for at least 12 months. Although Sulfamethoxazole-Trimethoprim and Linezolid have good CNS penetration, their well-known bone marrow toxicity makes prolonged treatment very challenging. In our case, immunodeficiency screening detected autoantibodies against GM-CSF as a possible cause of his disseminated nocardiosis with CNS involvement [5]. Anti-GM-CSF autoantibodies are a key risk factor and are increasingly recognized in adults with unexplained nocardiosis [6]. Additional risk factors for this patient included occupational exposure in construction through inhalation of contaminated soil and dust or possible direct skin contact with contaminated water [5].
Conclusions: This case highlights the importance of screening for immunodeficiency and considering anti-cytokine autoimmunity in the differential diagnosis of severe opportunistic infections, such as nocardiosis, in otherwise immunocompetent individuals.