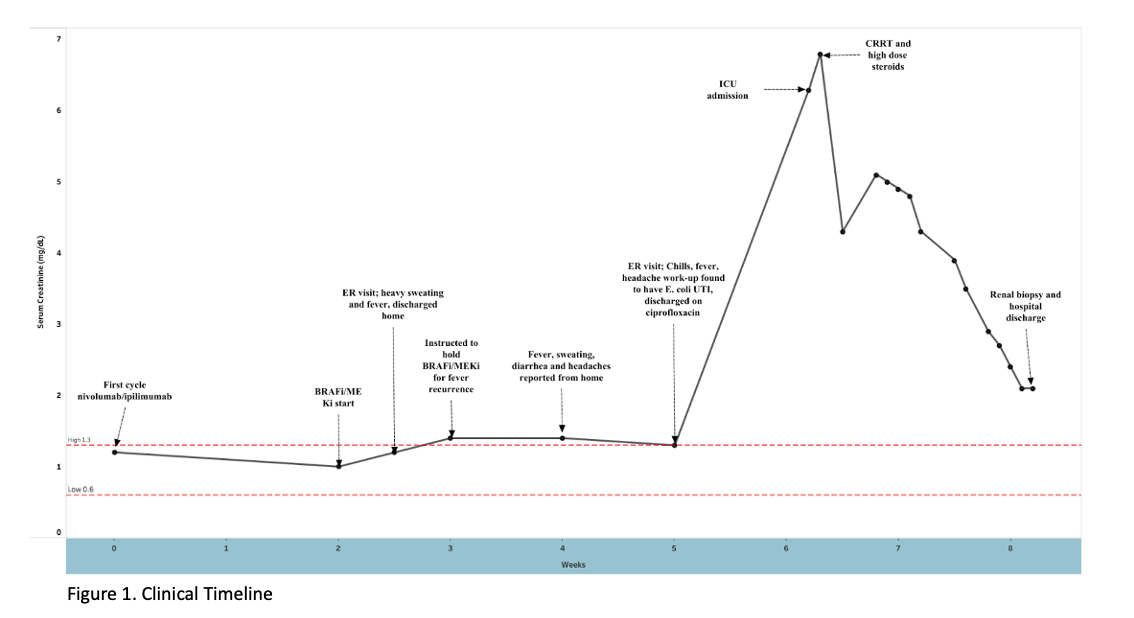
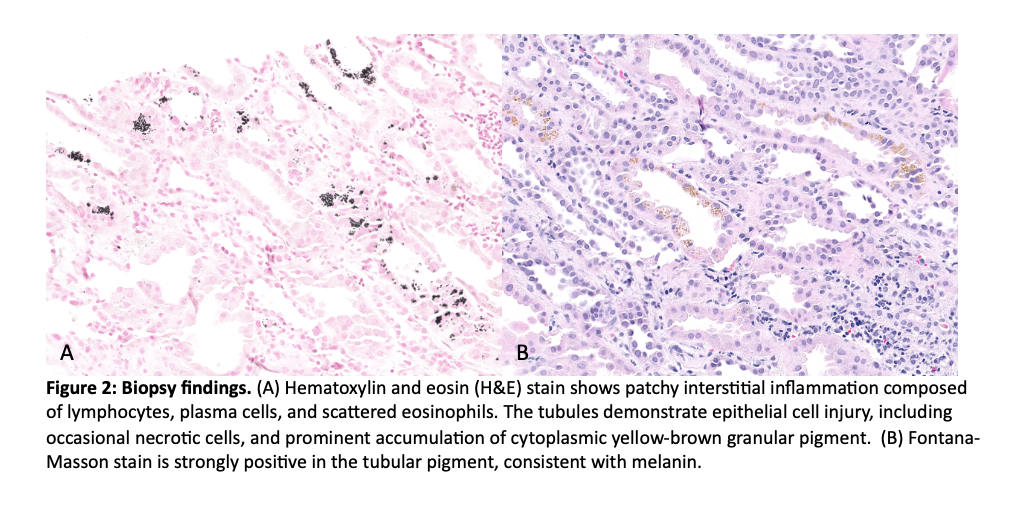
Case Presentation: This case investigates the etiology of acute interstitial nephritis (AIN) in a metastatic melanoma patient who was transitioned from immune-checkpoint inhibitors (ICIs) to targeted BRAF-MEK inhibitors (BRAFi/MEKi). A 73-year-old man with cutaneous melanoma with lung metastases presented to the emergency room for generalized weakness, fevers, and renal failure. Six weeks prior to his presentation, he started immunotherapy with nivolumab 1 mg/kg plus ipilimumab 3 mg/kg while awaiting results of somatic mutation testing. Two weeks after starting immunotherapy, genetic testing revealed positive BRAF V600E mutation and he was transitioned to oral BRAFi/MEKi, dabrafenib 150 mg twice daily and trametinib 2 mg daily. Within one week of starting BRAFi/MEKi, he reported intermittent fevers and diarrhea. His clinical symptoms worsened over the course of one month despite multiple ER visits (Fig. 1). Upon admission, labs were notable for severe thrombocytopenia (platelets 45 K/UL) and an increase in creatinine to 6.3 mg/dL (baseline 1.3 mg/dL) (Table 1). CT Chest demonstrated a new area of irregular dense airspace opacity in the left lower lobe, concerning for possible pneumonia. The patient was initiated on intravenous antibiotics for presumed sepsis and admitted to the intensive care unit. However, his kidney function failed to improve with fluids and he was started on continuous renal replacement therapy (CRRT) as well as intravenous methylprednisolone 500 mg. Blood, sputum, and urine cultures showed no growth. Two days later, his cytopenias and renal function improved enough to discontinue dialysis. A renal biopsy was performed and acute interstitial nephritis, most likely medication-induced. Ultimately, the patient was discharged home in stable condition with a prednisone taper.
Discussion: Few case reports describe either kidney immune-related adverse events (irAEs) for ICI or BRAF combination therapies. Identifying the culprit medication is important for determining future therapeutic options, particularly if intending to retrial the offending agent. Both ICI and BRAFi/MEKi-related kidney injuries are uncommon, with ICI-related kidney injuries ranging from 1.17-4.9% and dabrafenib-induced AIN reported at < 10% (trametinib not reported)(1-4). However, rates of kidney injury related to dabrafenib and trametinib are unclear, with one recent large retrospective study reporting that 21% of patients developed an AKI after exposure to dabrafenib(5). Although ICI-related AIN is better recognized, the close timing between administration of both medications and development of symptoms after initiation of BRAFi/MEKi makes it difficult to rule out BRAFi/MEKi as the possible culprit medication regimen. If a retrial of therapy is needed, more evidence exists to support the safety of retrial of ICI therapy, with recurrence rates reported at 12-20%(1,6). Limited data exists related to retrial of BRAFi/MEKi after treatment-related kidney injury and no specific guidelines exist(7).
Conclusions: In patients exposed to ICI and/or BRAFi/MEKi therapies, development of renal failure should prompt consideration of medication-related acute interstitial nephritis. Per NCCN guidelines, treating physicians should consider a kidney biopsy when severe toxicity occurs, namely a serum creatinine at least 4.0 mg/dL8,9. Although treatment of AIN is similar regardless of the specific inciting agent, recognition of the culprit medication when possible may guide future treatment decisions.