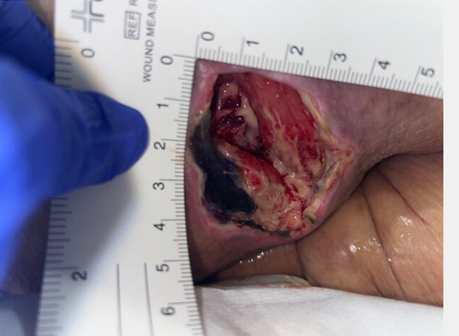
Case Presentation: A 74-year-old woman with stage 3 chronic kidney disease, chronic right hip prosthetic joint infection (PJI), hypertension, and bilateral hip arthroplasties was admitted for altered mental status, worsening renal function, and metabolic disturbances. She had a history of total femoral and knee arthroplasty for PJI, complicated by necrotic wounds and ongoing seropurulent drainage.The patient developed an acute kidney injury on chronic kidney disease, attributed to acute tubular necrosis, sepsis, and prior IV vancomycin exposure. Creatinine increased to 3.16 mg/dL and BUN to 105 mg/dL, necessitating intermittent hemodialysis and subsequent continuous renal replacement therapy, without anticoagulation due to bleeding risk.Blood cultures drawn grew Candida auris. Suspected infection sources included an infected right total hip arthroplasty and central venous catheter. The CVC was removed, and she was started on micafungin, meropenem, and daptomycin. Despite therapy, blood cultures remained persistently positive, raising concern for deep-seated infection. Transesophageal echocardiography was negative for endocarditis. Surgical planning for debridement of infected prosthetic tissue was planned. Additional complications included bilateral pleural effusions secondary to volume overload in the setting of acute renal failure, acute encephalopathy, anemia of CKD, and resolving pancreatitis.
Discussion: This case highlights the multifaceted challenges in treating Candida auris fungemia, especially in a patient with CKD and chronic PJI. C. auris is notoriously resistant to multiple antifungals—nearly universally resistant to fluconazole and increasingly to echinocandins (~8%), limiting treatment options.Persistent fungemia despite appropriate antifungal therapy and catheter removal suggests ongoing seeding from infected prosthetic material. Longstanding PJI and complex orthopedic hardware make source control difficult, contributing to treatment failure. In such settings, early surgical intervention, prolonged antifungal therapy, and multidisciplinary coordination are critical.Environmental persistence and frequent misidentification of C. auris increase the risk of nosocomial transmission. Rigorous infection control, including contact precautions and sporicidal disinfection, is essential. Diagnostic advances such as mass spectrometry and PCR enhance early detection but remain unavailable in many settings, reflecting broader disparities in fungal diagnostics.Her extensive surgical history placed her at high risk for persistent infection. Management of PJI requires combined medical and surgical strategies, particularly when C. auris is involved, given its propensity for biofilm formation and poor response to systemic therapy alone.
Conclusions: This case illustrates the clinical and therapeutic complexity of managing persistent Candida auris fungemia in a critically ill patient with CKD and chronic prosthetic infection. Successful outcomes require early recognition, echinocandin-based therapy, aggressive source control, renal-protective strategies, and robust infection prevention protocols. As C. auris continues to emerge globally, clinicians must maintain high clinical suspicion, utilize rapid diagnostics where available, and adopt a multidisciplinary approach to reduce mortality and transmission.