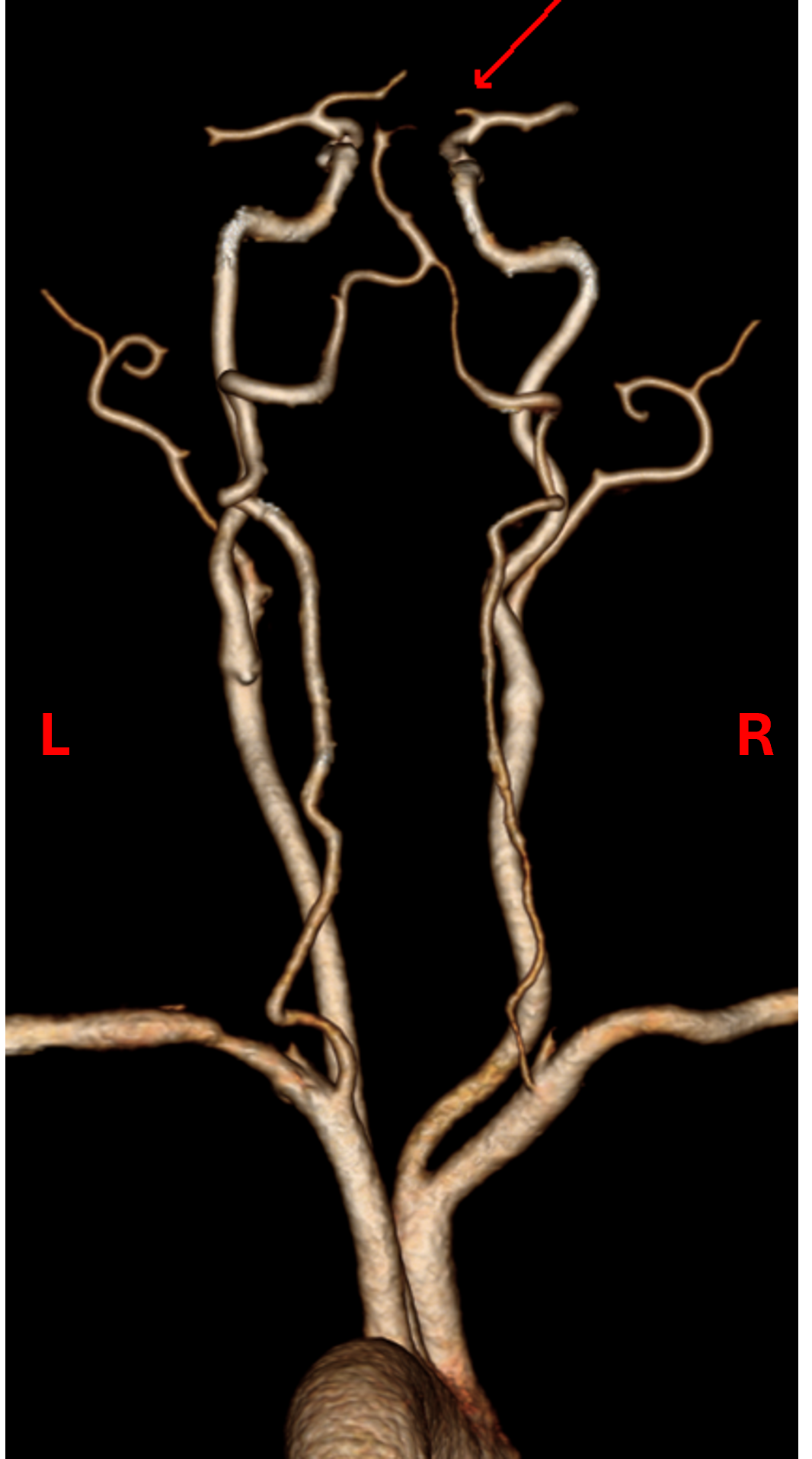
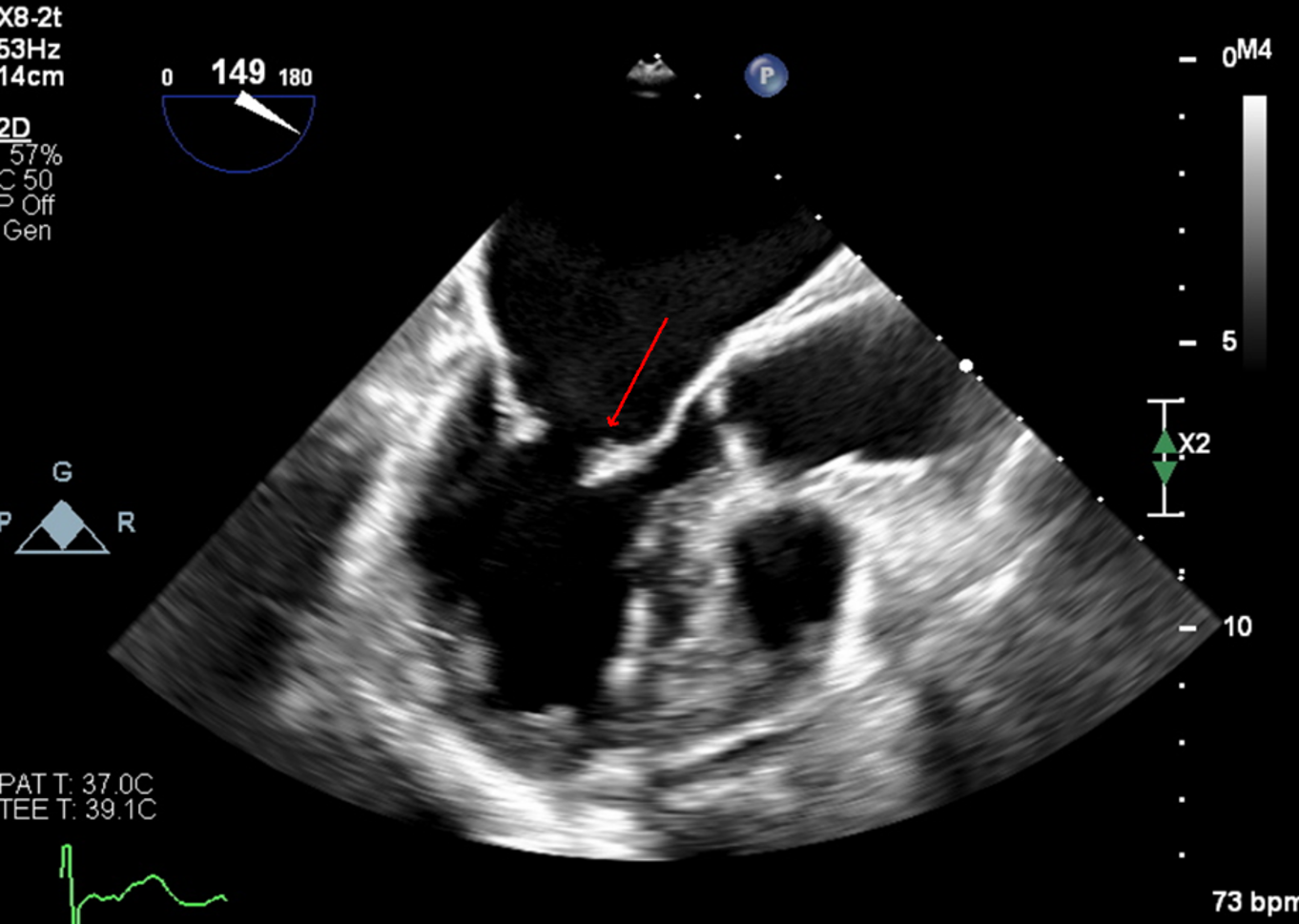
Case Presentation: A 67 year-old female presented to the ED with acute left upper extremity paresis and facial droop. She had 2 previous cardioembolic strokes, in 2007 from mitral nonbacterial thrombotic endocarditis for which she started warfarin and in 2010 after stopping warfarin for 8 months due to menorrhagia. Warfarin was switched to apixaban in 2021 due to patient preference. She also had atrial fibrillation (AF) with CHA2DS2-VASc 5 (diagnosed 2020), mild-moderate mitral regurgitation (on 2020 echocardiogram), hypertension, hyperlipidemia and a 37 pack-year smoking history.She was hypertensive (BP 172/89) with otherwise normal vital signs. Examination was significant for NIHSS 2 (left upper extremity drift and facial droop) and an irregularly irregular pulse. EKG confirmed AF. CT head and neck angiogram showed a proximal right M2 occlusion (Fig.1) with distal flow reconstitution and patent carotid arteries.Neurology, Vascular medicine and Cardiology teams were consulted. She was not a thrombolysis candidate due to apixaban intake and mild signs/symptoms. Apixaban peak and trough levels were both within the expected therapeutic range with anti-Xa levels of 130 and 77 respectively. Thrombophilia testing was negative for protein C, prothrombin gene mutation, factor V Leiden and antiphospholipid antibodies. Work-up for malignancy with CT imaging and cancer screening was unremarkable.Transesophageal echocardiogram was obtained to assess for a cardioembolic cause of stroke and revealed a hockey-stick appearance of the anterior mitral leaflet which is classic for rheumatic valve disease (Fig.2), moderate mitral stenosis and moderate mitral regurgitation. Ejection fraction was preserved at 50% and there was no intracardiac mass or patent foramen ovale.Apixaban had been inadequate anticoagulation as she now had both moderate rheumatic mitral stenosis and atrial fibrillation. She was bridged to warfarin with target INR 2-3 and discharged to valve clinic for follow-up.
Discussion: Stroke causes considerable morbidity and mortality as the 5th leading cause of death in the USA (1) and comprises ∼1 in 20 ED visits that result in hospitalizations (2). Our patient’s stroke was cardioembolic due to ‘valvular AF’ (new moderate rheumatic mitral stenosis with AF) for which warfarin therapy is superior to DOAC for stroke prevention (3). Rheumatic mitral stenosis is challenging to diagnose for its rarity and insidious presentation decades after rheumatic fever, which itself is underdiagnosed (4). Indeed, our patient had no history of rheumatic fever or new cardiac signs/symptoms. Early treatment of Group A Streptococcus infection and secondary prevention of rheumatic fever with penicillin is crucial to prevent permanent valve damage that occurs in rheumatic heart disease (4).
Conclusions: We illustrate a rare case of recurrent ischemic stroke due to DOAC failure in our patient with AF and new rheumatic mitral stenosis without a known history of rheumatic fever. Patients taking a DOAC who present with ischemic stroke of suspected cardioembolic etiology should be evaluated for medication compliance, hypercoagulability and intracardiac pathology. It is key to maintain a high index of clinical suspicion for ‘Valvular AF’, especially in those with known AF, as it can present insidiously and requires warfarin for optimal stroke prevention.