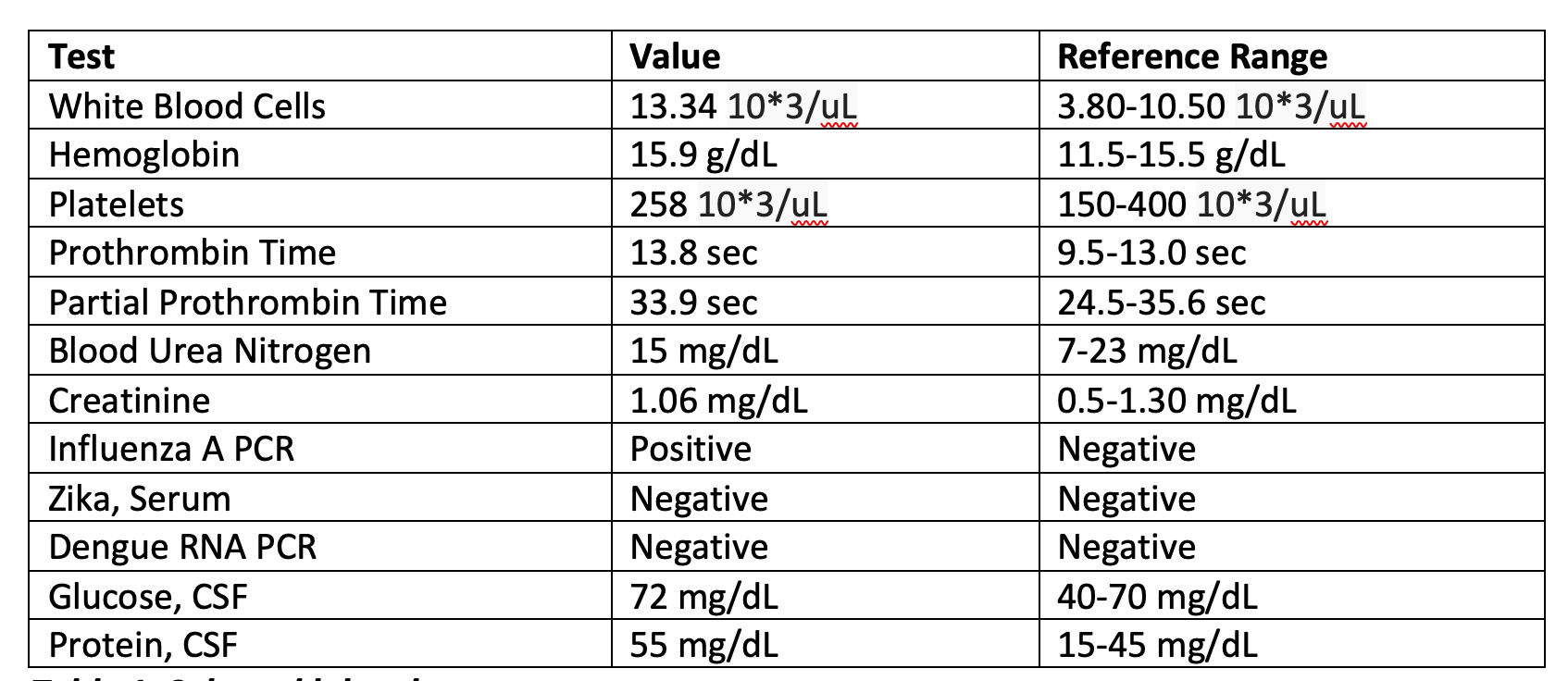
Case Presentation: A 60-year-old female with type 2 diabetes, depression, and hyperlipidemia presented with a week of nausea, vomiting, abdominal discomfort, and fatigue. She denied fever, diarrhea, chest pain, or dyspnea. Since starting Tirzepatide 7.5 mg weekly three weeks prior, she had reduced oral intake and unintentional weight loss. Her diabetes was previously managed with metformin 500 mg daily and Dulaglutide, without insulin use. She denied alcohol use, infection, or other medication changes.On exam, she was alert but fatigued, tachycardic (HR 125 bpm), mildly dehydrated, BP 142/90 mmHg, RR 18, afebrile, and saturating 95% on room air. Laboratory results showed glucose 139 mg/dL, bicarbonate 17 mmol/L, anion gap 25 mmol/L, β-hydroxybutyrate 5.3 mmol/L, arterial pH 7.29, and mild hyponatremia (Na 134 mmol/L). Urinalysis was positive for ketones and leukocyte esterase but negative for nitrites.Findings were consistent with euglycemic diabetic ketoacidosis (euDKA). She was treated with intravenous fluids, insulin infusion, and electrolyte monitoring per DKA protocol. Tirzepatide was discontinued. Within 48 hours, her metabolic parameters normalized, the anion gap closed, and she was transitioned to subcutaneous insulin. She was discharged in stable condition with instructions to avoid GLP-1/GIP agonists and follow up with endocrinology for reassessment of diabetes therapy.
Discussion: Euglycemic diabetic ketoacidosis (euDKA) is a rare, potentially life-threatening DKA variant marked by ketosis and metabolic acidosis with normal or mildly elevated glucose (< 250 mg/dL). Representing 2–10% of DKA cases, it poses a diagnostic challenge as the absence of hyperglycemia may delay recognition. While SGLT2 inhibitors are the usual culprits, this case highlights a possible association with Tirzepatide (Mounjaro), a dual GLP-1/GIP receptor agonist approved in 2022, with few reports linking it to euDKA.In euDKA, normal glucose levels arise from reduced oral intake, enhanced glucagon-driven ketogenesis, and suppressed hepatic glucose output. Tirzepatide’s common side effects including nausea, vomiting, and appetite suppression can cause caloric restriction and relative insulin deficiency, promoting ketosis. Transitioning off insulin or concurrent dehydration may heighten risk.Clinicians should suspect euDKA in Tirzepatide users presenting with nausea, vomiting, or abdominal pain even when glucose < 250 mg/dL. Diagnosis relies on serum or urine ketones, β-hydroxybutyrate, pH, and anion gap assessment. Management parallels classic DKA with IV fluids, insulin infusion, and electrolyte monitoring with earlier dextrose use to prevent hypoglycemia.This case underscores the need for patient counseling on DKA symptoms, avoidance of excessive fasting, and close monitoring after Tirzepatide initiation. Further research is needed to clarify this emerging association.
Conclusions: This case illustrates a rare but important potential adverse effect of Tirzepatide: euglycemic diabetic ketoacidosis. As the use of GLP-1/GIP receptor agonists expands for diabetes and weight management, clinicians should be aware of this presentation and consider euDKA in any patient on Tirzepatide presenting with unexplained gastrointestinal or constitutional symptoms, even in the absence of significant hyperglycemia.