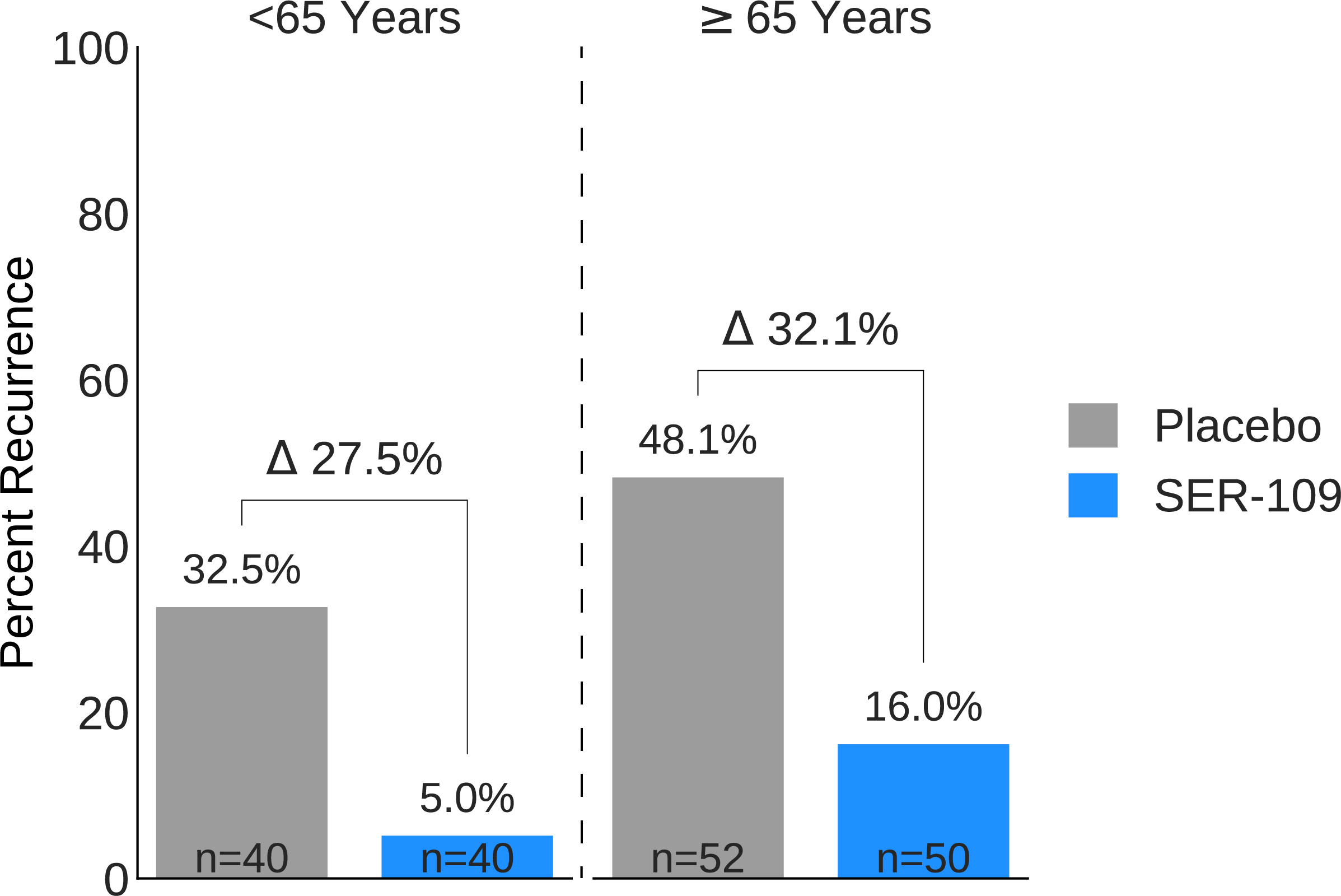Background: Antibiotics targeted against C. difficile bacteria are necessary, but insufficient, to achieve a durable clinical response because they have no effect on C. difficile spores that germinate within a disrupted microbiome. ECOSPOR-III evaluated SER-109, an investigational, biologically-derived microbiome therapeutic of purified Firmicute spores for treatment of recurrent CDI.
Methods: Adults ≥18 years with recurrent CDI (≥3 episodes in 12 months) were screened at 75 US/CAN sites. CDI was defined as ≥3 unformed stools/day for ≥48 hours with a (+) C. difficile assay. After completion of 10-21 days of vancomycin or fidaxomicin, adults with symptom resolution were randomized 1:1 to SER-109 (4 capsules x 3 days) or matching placebo and stratified by age (≥ or <65 years) and antibiotic received. Primary objectives were safety and efficacy at 8 weeks. Primary efficacy endpoint was recurrent CDI (recurrent toxin+ diarrhea requiring treatment); secondary endpoints included efficacy at 12 weeks after dosing.
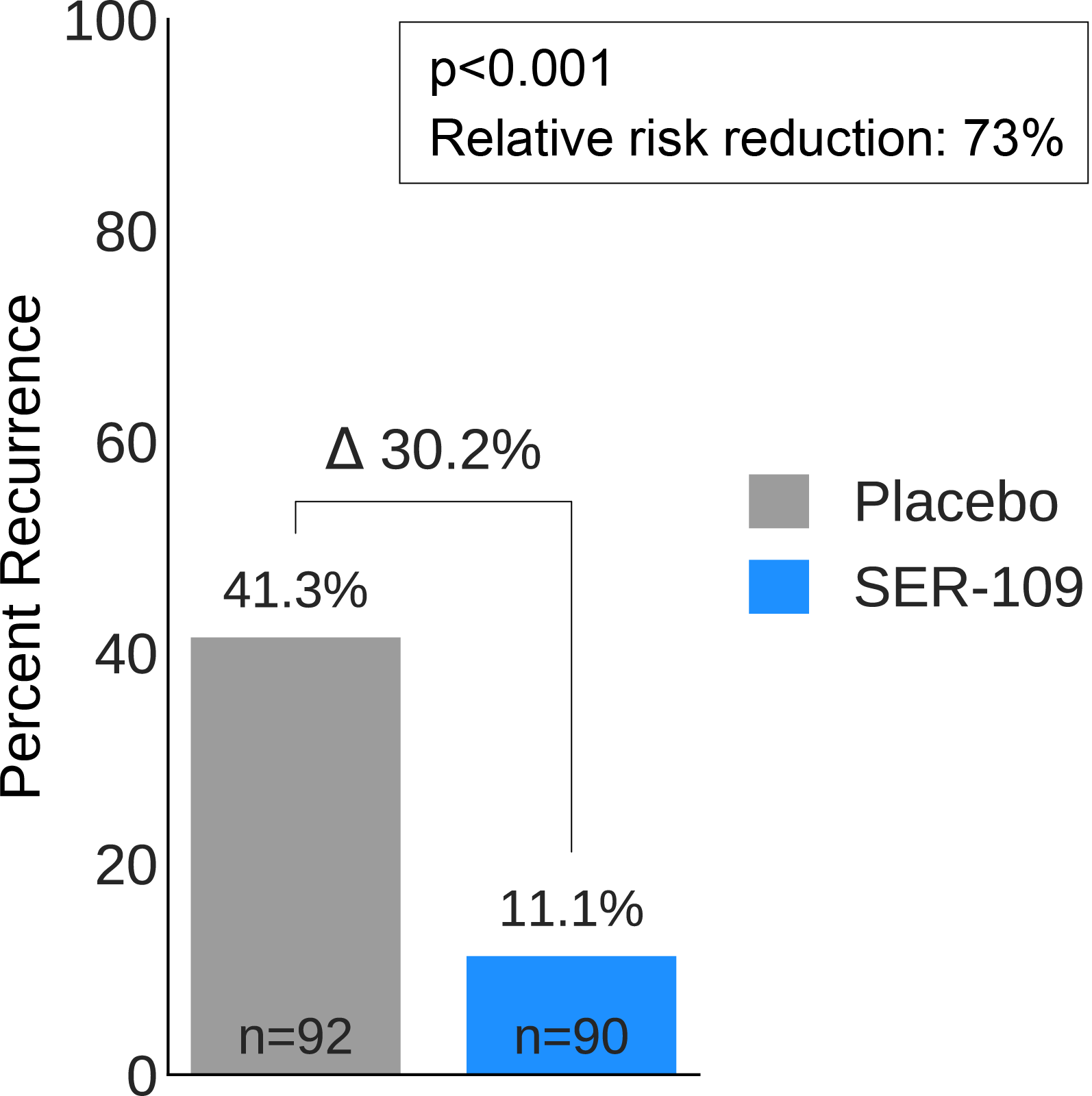
Results: 287 subjects were screened and 182 randomized (59.9% female; mean age 65.5 years). The most common reason for screen failure was a negative C. difficile toxin assay. A significantly lower proportion of SER-109 subjects had recurrent CDI after dosing compared to placebo at week 8 (11.1% vs 41.3%, respectively; RR 0.27; 95% CI 0.15, 0.51; p-value <0.001; Figure 1). This translates into a higher rate of sustained clinical response with SER-109 compared to placebo (88.9% vs 58.7%, respectively). Similarly, a significantly lower proportion of SER-109 subjects had recurrent CDI compared to placebo at week 12 (16.7% vs 47.8%, respectively; RR 0.35; 95% CI 0.21, 0.58; p-value <0.001). Efficacy rates were significantly higher with SER-109 vs placebo in both stratified age groups (Figure 1). SER-109 was well-tolerated with a safety profile similar to placebo. The most common treatment-emergent adverse events (TEAEs) were gastrointestinal and were mainly mild to moderate. No serious TEAEs, infections, deaths or drug discontinuations were deemed related to study drug.
Conclusions: SER-109, an oral live microbiome therapeutic, achieved high rates of sustained clinical response with a favorable safety profile. By enriching for Firmicute spores, SER-109 achieves high efficacy, while mitigating risk of transmitting infectious agents, beyond donor screening alone. SER-109 represents a major paradigm shift in the clinical management of patients with recurrent CDI.