Background: Certain infections, such as mucormycosis and Fournier gangrene, were initially linked to diabetes via case reports (1). Subsequent studies using electronic health record (EHR) data and frequentist methods have provided new insights, including increased mortality among patients with diabetes and infections (2). However, infection risk among patients with type 1 diabetes (T1D), type 2 diabetes (T2D), and prediabetes remains poorly characterized compared to other diabetes-related complications.
Methods: We developed a probabilistic graphical model (PGM) to characterize diverse infection outcomes using a retrospective cohort study design. Adults aged 18 years or older with a prior diagnosis of T1D (n = 9,476), T2D (n = 74,270), and prediabetes (n = 32,095) from the University of Utah’s EHR database were included. Comparator groups included patients without diabetes (n = 1,373,245) and the general population (n = 1,479,638). Using the PGM, we computed risk ratios (RR) and 95% confidence intervals (CI) for composite infection outcomes organized by organ system, pathogen type, and severity. We then computed RR for 1,356 individual infection outcomes, defined by diagnostic (i.e., ICD) and laboratory (i.e., LOINC) codes, and explored effects of social determinants of health.
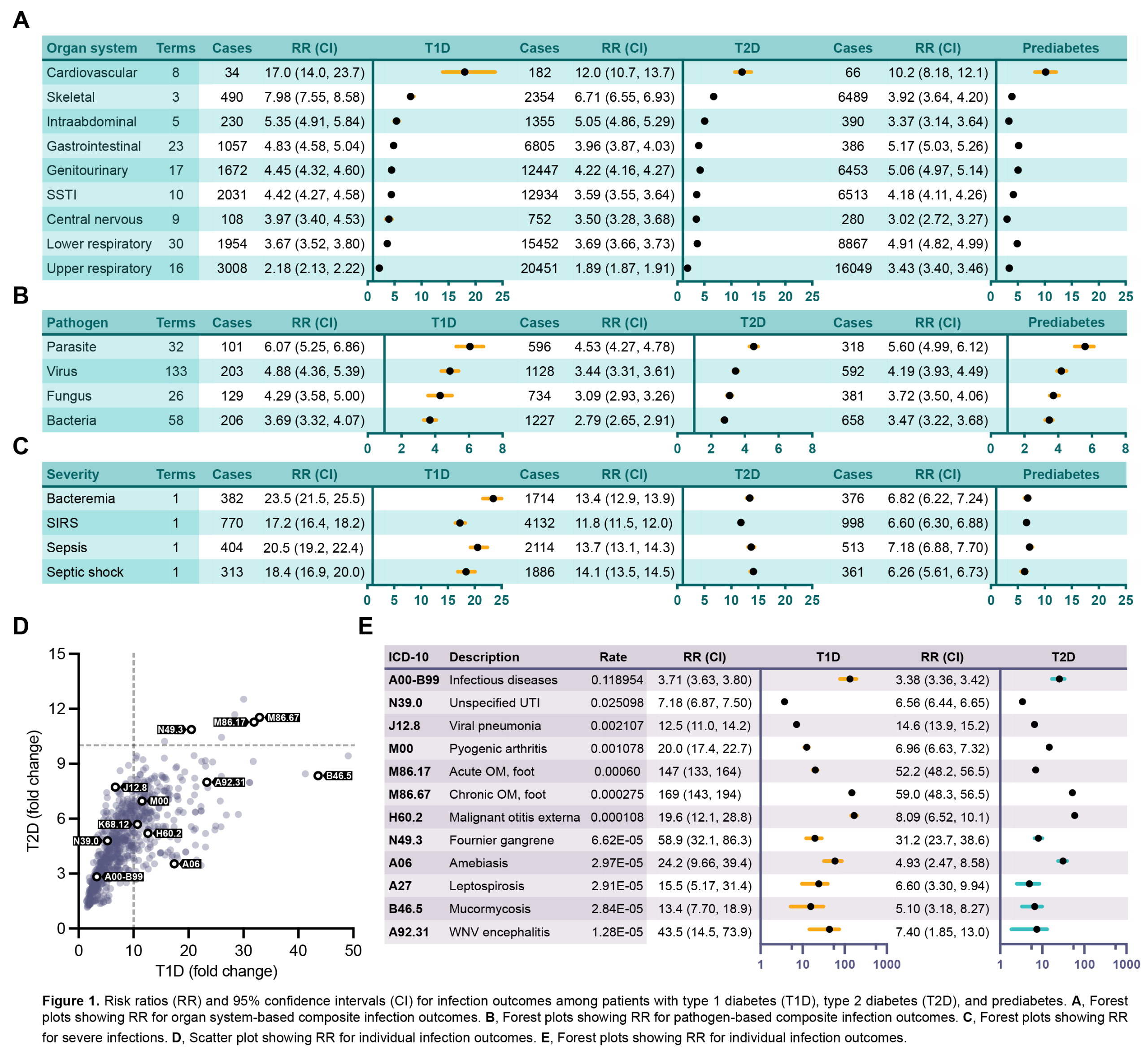
Results: Compared to those without diabetes, patients with T1D, T2D, and prediabetes had multifold increased risk for all composite infection outcomes. Risk was greatest for the cardiovascular infection outcome among patients with T1D (RR = 17.0, CI 14.0–23.7), T2D (RR = 12.0, CI 10.7–13.7), and prediabetes (RR = 10.2, CI 8.18–12.1). Likewise, risk was greatest for the parasitic infection outcome among patients with T1D (RR = 6.07, CI 5.25–6.86), T2D (RR = 4.53, CI 4.27–4.78), and prediabetes (RR = 5.60, CI 4.99–6.12). The T1D cohort had the greatest fold-change risk for severe infections such as bacteremia (RR = 23.5, CI 21.5–25.5), systemic inflammatory response syndrome (RR = 17.2, CI 16.4–18.2), sepsis (RR = 20.5, CI 19.2–22.4), and septic shock (RR = 18.4, CI 16.9–20.0).Compared to the general population, patients with T1D, T2D, and prediabetes had increased risk for most individual infection outcomes. Of 1,356 ICD and LOINC terms, we found significantly increased RR for 878 (64.75%) in T1D, 1,047 (77.21%) in T2D, and 989 (72.94%) in prediabetes. As expected, risk was increased for established diabetes-associated infections such as mucormycosis (RR = 13.4, CI 7.70–18.9 for T1D; RR = 5.10, CI 3.18–8.27 for T2D) and Fournier gangrene (RR = 58.9, CI 32.1–86.3 for T1D; RR = 31.2, CI 23.7–38.6 for T2D). However, we also identified novel associations such as West Nile Virus encephalitis (RR = 43.5, CI 14.5–73.9 for T1D; RR = 7.40, CI 1.85–13.0 for T2D) and leptospirosis (RR = 15.5, CI 5.17–31.4 for T1D; RR = 6.6, CI 3.18–8.27 for T2D). Age, sex, ethnicity, ancestry, and insurance status modified but did not fully explain the increased risk.
Conclusions: Infection outcomes are complex and thus difficult to characterize. Using a robust PGM-based approach, we identified multifold increased risk for diverse infection outcomes for patients with T1D, T2D, and prediabetes. Our findings highlight a need to better predict and prevent infections among patients with diabetes.