Background: An average of 10% of patients in the U.S. report having a penicillin allergy yet the Centers for Disease Control estimates that less than 1% of the population are truly allergic. It is well described in adult literature that broad spectrum antibiotics are often used as penicillin alternatives and this may contribute to negative downstream effects such as suboptimal therapy, increased risk for antibiotic resistance, and higher costs. There is a paucity of literature in the inpatient pediatric population as to the impact of a penicillin allergy label. Study objectives included estimating the rates of penicillin-class allergies in our pediatric inpatients, comparing demographics, and evaluating antibiotic use in inpatients with and without penicillin allergy labels.
Methods: This study was a retrospective chart review of pediatric patients admitted to a large free-standing children’s hospital in 2019. Included were all floor-status inpatients aged 0-18 years. Excluded were patients with immunocompromised or organ transplant status and those admitted to the ICUs or the oncology service, due to potential for multiple confounders impacting antibiotic prescribing practices. Data extracted from the electronic medical record identified patients admitted to the hospital and stratified them by penicillin allergy status. Penicillin-class allergy was defined as allergy to penicillin, amoxicillin or ampicillin. Antibiotics prescribed during hospitalizations were electronically extracted but categorized into drug classes manually. Differences in demographics and antibiotic prescribing practices between inpatients with and without penicillin-class allergies were examined using non-parametric analyses (Fisher exact, Wilcoxon rank sum test), as appropriate for the data distribution.
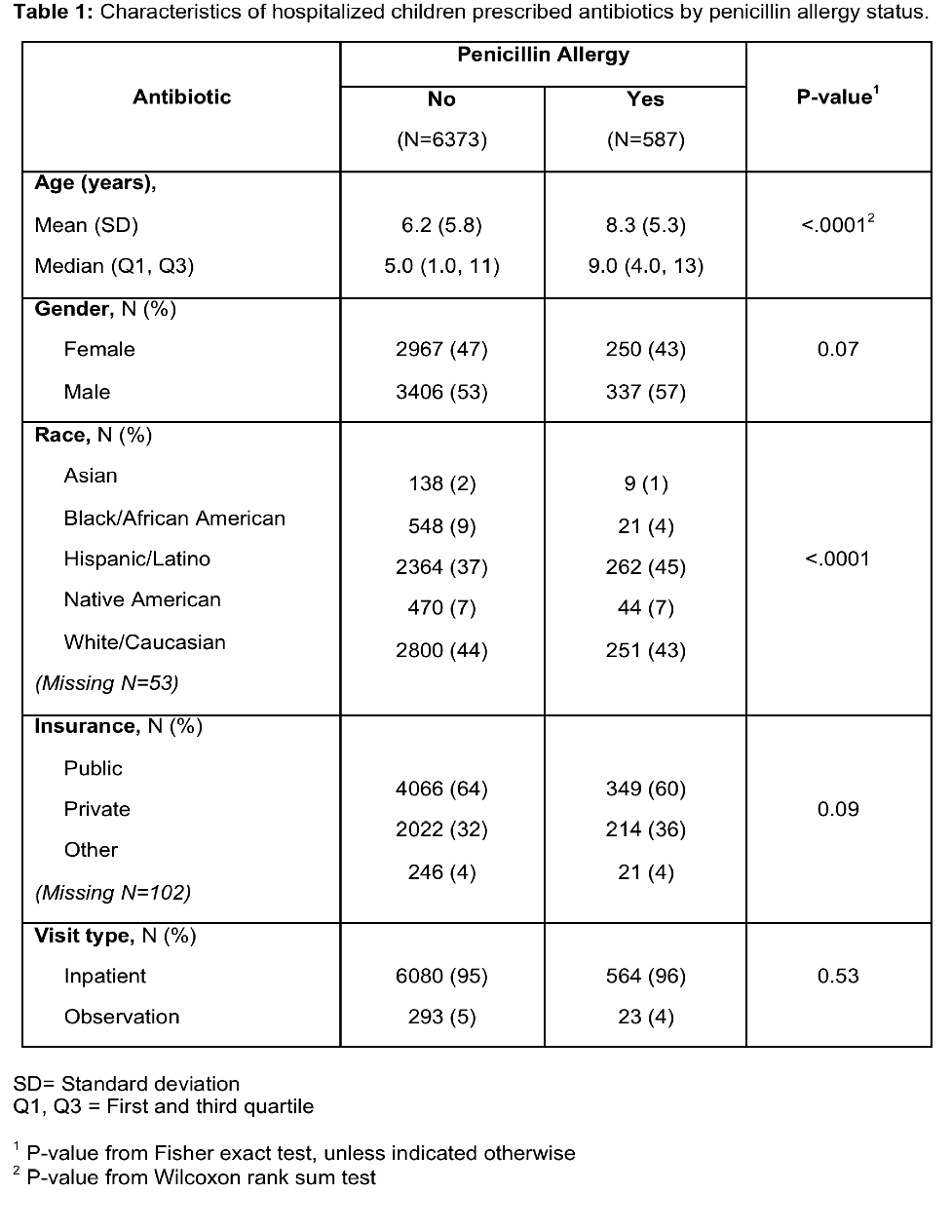
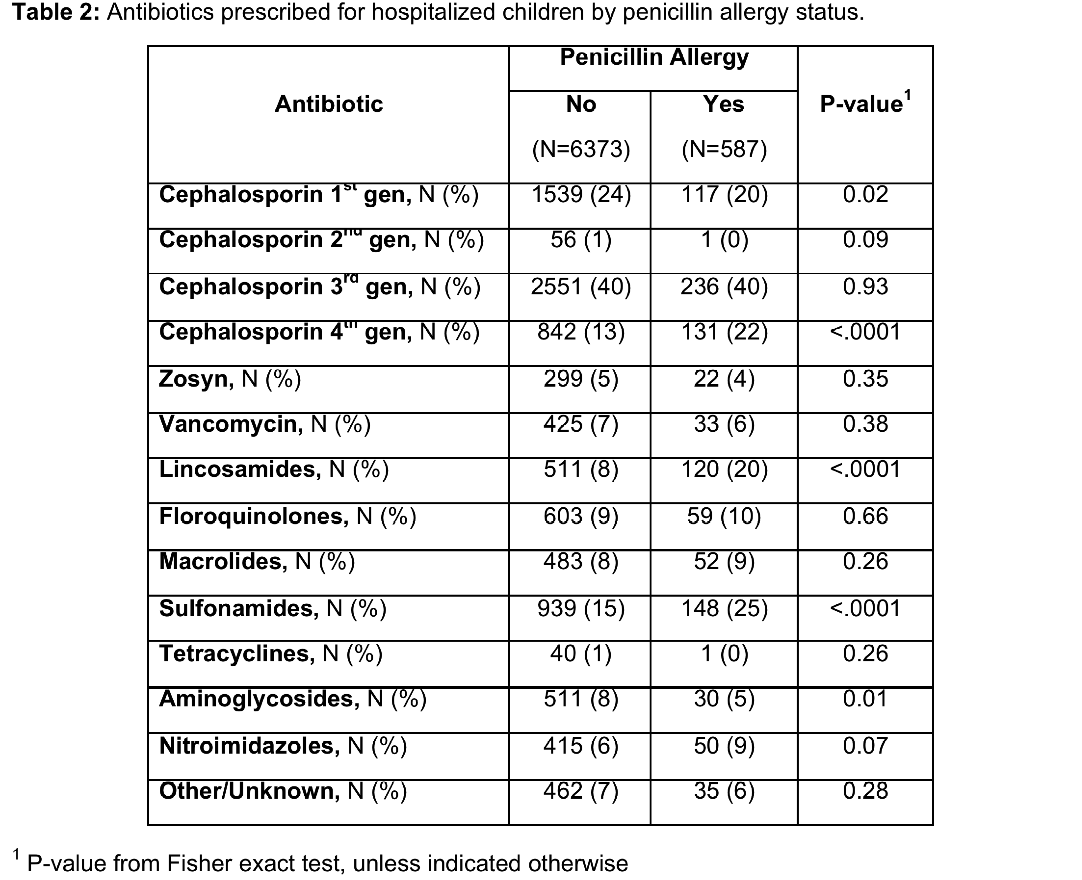
Results: Overall, 13,273 patients met inclusion criteria and 1,139 (8.6%) had a penicillin allergy label. A total of 6,960 inpatients received antibiotics during their hospitalization, and were included in data analysis. Of those patients with a penicillin allergy label, 587 (51.5%) received antibiotics during their stay. A total of 6,373 (52.5%) patients without the allergy received an antibiotic during their stay. Allergic patients prescribed antibiotics were older (mean age 8.3 vs. 6.2 years, p-value <.0001) and more likely to be Hispanic/Latino (45% vs 37%, p-value <.0001) (Table 1). Significantly higher antibiotic prescribing rates were found in patients with a penicillin allergy label as compared to non-allergic patients for the 4th generation cephalosporins (22% vs 13%, p-value <.0001), lincosamides (20% vs 8%, p-value <.0001) and sulfonamides (25% vs 15%, p-value <.0001). In contrast, prescribing rates for 1st generation cephalosporins (20% vs 24%, p-value 0.02) and aminoglycoside (5% vs 8%, p-value 0.01) were significantly lower in penicillin allergic vs. non-allergic patients (Table 2).
Conclusions: The burden of patients with reported penicillin allergies at our institution was 8.6% of inpatients, similar to literature estimates. Patients with penicillin allergies were older. There was increased use of 4th generation cephalosporin, licosamide, and sulfonamide antibiotic classes in patients with a penicillin allergy label. Our findings suggests a higher percentage of patients with a reported penicillin allergy receive broad spectrum antibiotics which has potential risks, highlighting the importance of truly confirming patients’ allergy status and judicious antibiotic prescribing.