Background: Community acquired pneumonia (CAP) is a leading cause of hospitalization, economic burden and excess antibiotic use in the United States.1,2 Previously, a multidisciplinary group at University of Utah Health launched a clinical decision support triggered CAP pathway in 2017 that was associated with cost savings and decreased duration of antibiotics.3 Based on review of the literature and local epidemiology, antimicrobial stewardship and hospital medicine led updates to the existing CAP care pathway to focus on promoting appropriate laboratory utilization, narrowing empiric and definitive antibiotics, and shortening total duration of therapy for non-ICU patients with CAP. We aimed to assess the impact of our intervention on diagnostic and antibiotic treatment measures as well as associated clinical outcomes.
Methods: On 11/11/2021, the CAP pathway was updated for non-ICU patients to encourage narrow empiric antibiotics (i.e., ceftriaxone alone) without empiric atypical coverage for most patients, automatic de-escalation to oral amoxicillin on day 2, and a shortened total antibiotic duration of 3 days. The order set also emphasized appropriate, evidence-based diagnostic testing (i.e., no blood or respiratory cultures in patients without risk factors for resistant organisms). This retrospective, observational pre-post quality improvement study included non-ICU hospitalized adult patients with a discharge ICD10 code of pneumonia who received more than 1 day of antibiotics within 48 hours of admission. Patients with a hospital diagnosis of COVID were not included. Primary outcomes were antibiotic duration and azithromycin use. Secondary outcomes included composite 30-day readmission and 30-day mortality, amoxicillin use, length of stay (LOS) and ICU transfers. Outcomes were adjusted for age, CCI, and severity index (presence of >2: RR>20, HR>90, BP< 90mmHg, T >38 or < 35).
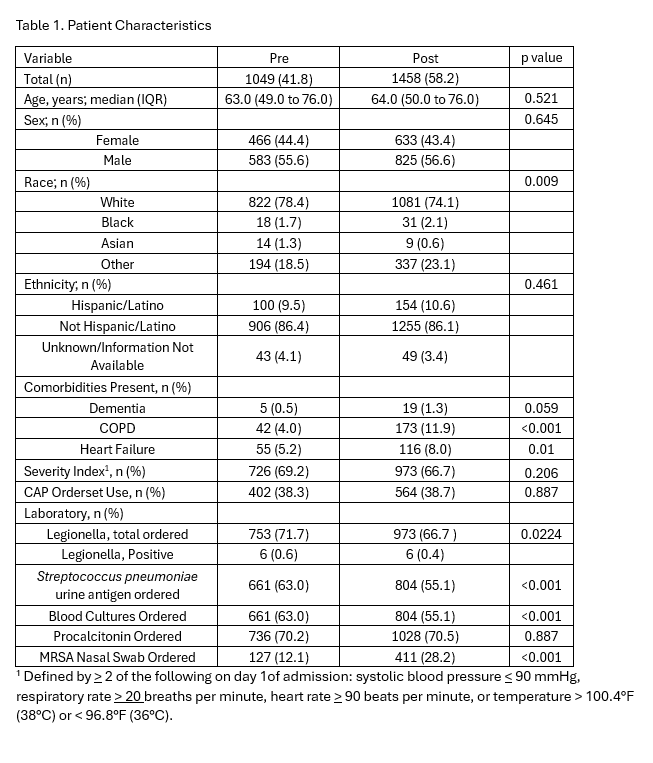
Results: A total of 1049 patients were included in the baseline period from 1/1/2019 -12/2021 and 1458 were included in the post implementation period of 1/2022 -12/2023 (Table 1). Demographic and clinical differences between baseline and post implementation were analyzed using the Mann–Whitney U test for continuous variables and the Chi-square test for categorical variables. Comorbidities including COPD and heart failure significantly increased in the post implementation population (p < 0.001, p =0.01) and 409 additional encounters were documented in the post implementation period consistent with hospital growth (Table 1). Both pre- and post-intervention, 38% of CAP admissions used the CAP orderset (Table 2). Azithromycin use decreased by 22.5% from baseline (p < 0.001) while median antibiotic duration remained unchanged (Table 2). Composite 30-day readmission and mortality as well as LOS were unchanged while floor to ICU transfers were lower (Table 2). Use of amoxicillin increased by 45.7% (p< 0.001, Table 2).
Conclusions: Updates to an existing CAP pathway led to anticipated reductions in azithromycin use and an increase in amoxicillin use without associated harm. Despite prompting from the order set, we did not see a decrease in antibiotic duration below our already low duration of 5 days. This is likely multifactorial and related to patients presenting with multiple comorbidities and providers not wanting to discontinue antibiotics before discharge. Further attempts to better understand the antibiotic discharge process and why attempts to shorten total antibiotic duration fail are needed.