Background: In our NIH funded multi-site cluster randomized clinical trial (RCT), we used implementation science methods to develop and implement quality improvement programs consisting of evidenced-based interventions to improve care transitions for patients hospitalized with chronic obstructive pulmonary disease (COPD). Since the hospitals were the ‘subject’ of study, typical patient-level RCT enrollment practices were not applicable. Therefore, in this pragmatic study, we developed step-wise enrollment and onboarding processes to ensure institutional support, ethical approval, and rigorous data use procedures. We obtained evaluations to understand how sites experienced this process.
Methods: Site recruitment ran September 2021 – December 2022 through the Hospital Medicine Reengineering Network (HOMERuN) and the Society of Hospital Medicine (SHM) via emails and newsletters sent to organizations’ listservs and SHM’s website. Enrollment was a three-step process: a site interest form, application form, and an enrollment process to ensure regulatory and data use agreements (DUAs) were secured and institutional support was assured: letters of support from organizational leaders, data officials, and site lead; ethical approval (IRB or QI determination); optional data use agreements (DUAs; all data de-identified). Onboarding sessions provided an overview of the study and outlined the expectations of the site leads. Site leads received an evaluation of the recruitment and enrollment process via REDCap to provide insights for improvements for future study enrollment.
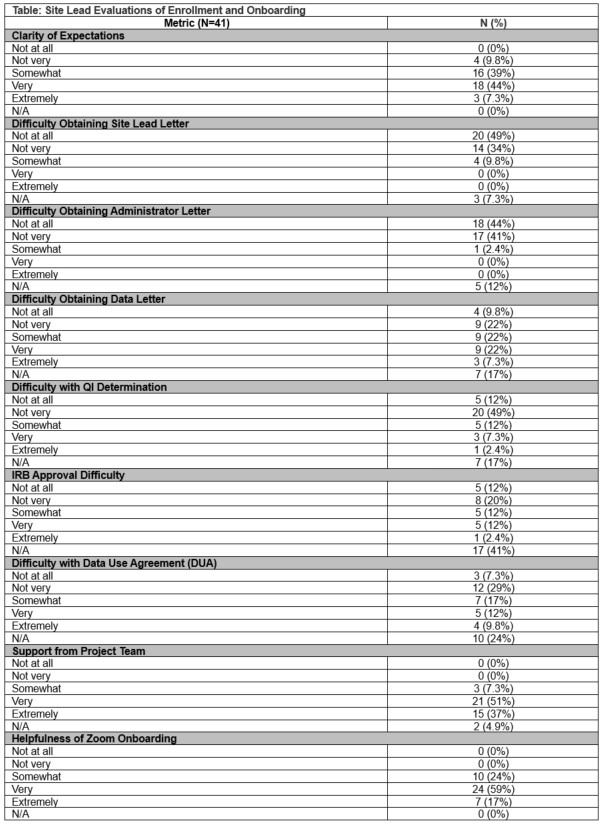
Results: Among 44 site leads across 21 sites, we received 41 completed evaluations (93%). Overall, 100% reported being supported during the enrollment process. Specifically, most (90%) reported being clear on expectations and few found obtaining the site lead (10%) or administrative (3%) letter of support difficult to obtain. However, the majority found obtaining the data administrator letter difficult (62%). The majority did not find it difficult to obtain IRB approval (54%), QI determination (74%), or DUAs (71%). Regarding onboarding, individual virtual onboarding sessions were held with each site and 100% of site leads reported these onboarding session to be helpful. (Table 1)
Conclusions: The site lead feedback provided critical insights into our study’s recruitment and enrollment. The results indicate that some sites had challenges in securing ethical approval and DUAs, and most found obtaining the data administrator letter of support difficult. However, the high level of satisfaction with the enrollment support and with the onboarding sessions highlights the strengths of our process. Further, the rigorous enrollment process may have led to increased engagement as only one site left the study after enrolling. The novel use of individual site onboarding was all well received. Overall, these findings underscore the importance of supporting the site-level enrollment and onboarding process.