Background: Hospital antibiotic stewardship programs (ASPs) play a critical role in minimizing unnecessary antibiotic use and combating antimicrobial resistance. Although various antibiotic stewardship interventions have been described, the feasibility and sustainability of these approaches remain uncertain. Furthermore, some interventions may be challenging for smaller hospitals to implement due to informatics resource limitations. Thus, we aimed to understand feasibility and sustainability of common antibiotic stewardship interventions and the relationship of sustainability with hospital bed size.
Methods: A cross-sectional survey was conducted between April and May 2023 across 69 hospitals in Michigan participating in the Michigan Hospital Medicine Safety Consortium, representing both large (≥200 beds) and small (< 200 beds) hospitals. Quality improvement or antimicrobial stewardship staff from each hospital ranked the feasibility of 7 common antibiotic stewardship interventions on a scale from 1 (easiest) to 7 (hardest). Respondents were then asked to report their status with 43 individual stewardship interventions as: a) implemented and sustained, b) implemented, but not sustained, c) tried but unable to implement, or d) never done. We used descriptive statistics and Fisher’s exact tests to compare reported intervention feasibility and implementation by hospital bed size (small vs. large).
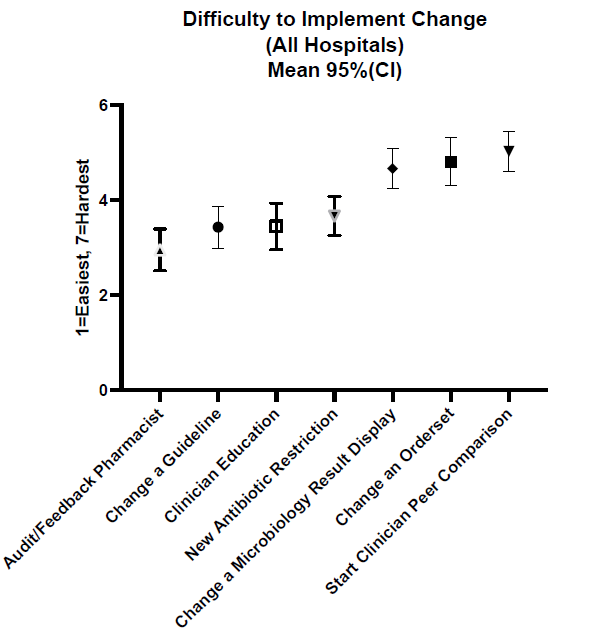
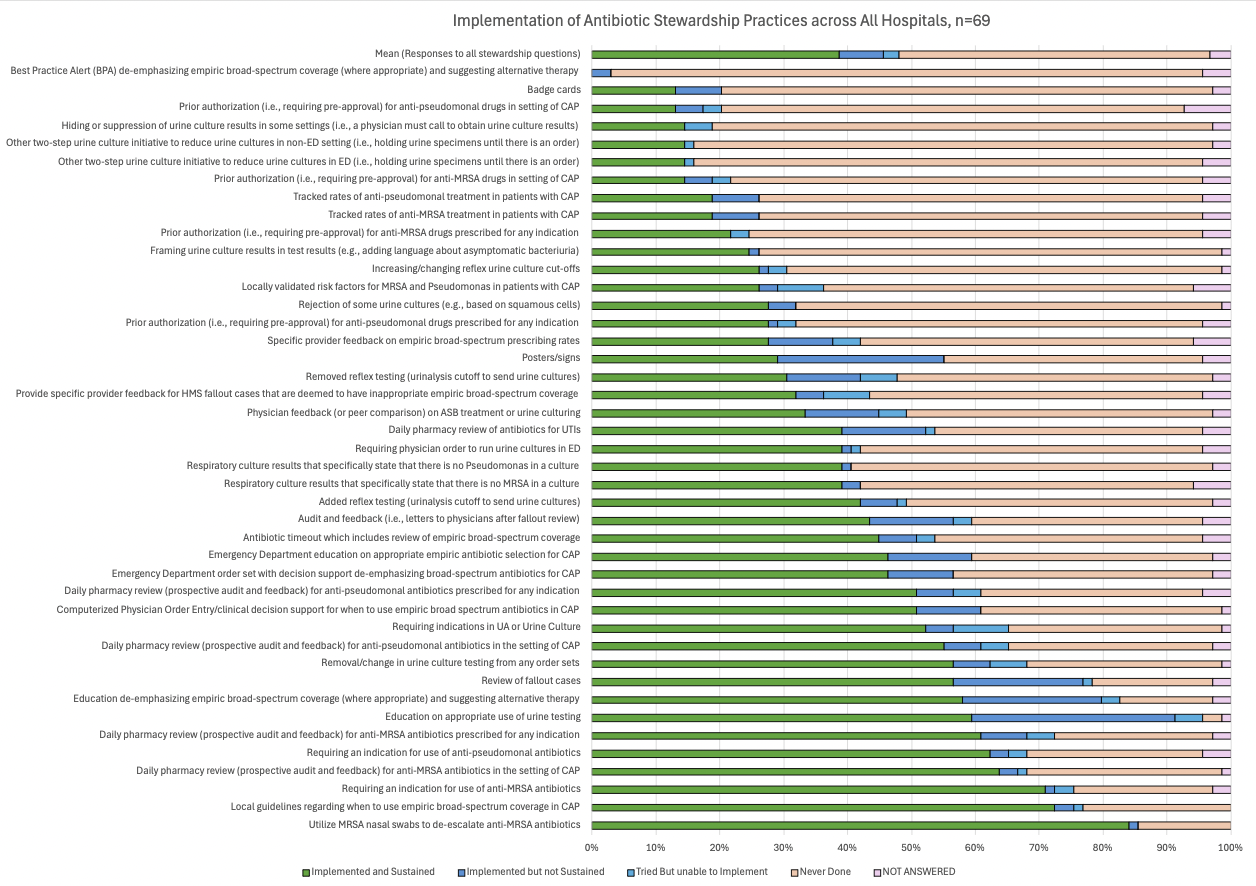
Results: All 69 hospitals responded to our survey (100% response rate). Across all hospitals, increasing audit and feedback by pharmacists was reported as the easiest new intervention to implement, whereas starting clinician peer comparison was reported as the hardest (Figure 1). Hospitals had implemented and sustained multiple stewardship interventions with substantial variation by intervention (Figure 2). Reported feasibility of the 7 common stewardship interventions did not significantly differ between large and small hospitals. However, small hospitals had significantly higher implementation of five antibiotic stewardship interventions: removal or change in order sets in urine culture testing (implemented by 73.1% of small hospitals vs. 46.3% of large hospitals; p=0.04), two-step urine culture initiative to reduce unnecessary testing (27% vs. 7%; p=0.04), Emergency Department order set with decision support de-emphasizing broad-spectrum antibiotics for CAP (77% vs. 48%; p = 0.02), daily pharmacy review of antibiotics for UTIs (58% vs. 30%; p=0.04), and daily pharmacy review of anti-pseudomonal antibiotics for CAP (73% vs. 46%; p=0.04).
Conclusions: Self-reported feasibility and implementation of antibiotic stewardship interventions varied widely across hospitals. However, once implemented, stewardship interventions were largely sustained—particularly those favoring technical (i.e., change in technology or process) over adaptive (i.e., require cultural adjustments) solutions. Hospital size influenced implementation for 5 of the 43 interventions. Small hospitals reported significantly higher rates of some antibiotic stewardship interventions potentially due to smaller patient populations and fewer administrative barriers to modifying existing practices. Hospitals should consider their local context, intervention feasibility and the potential for intervention sustainability when deciding on implementation priorities in stewardship.