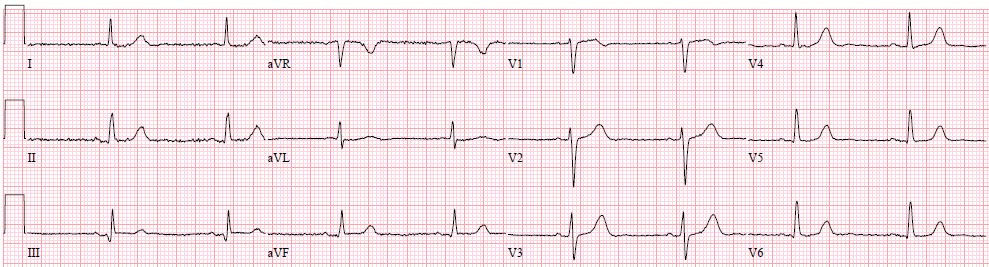
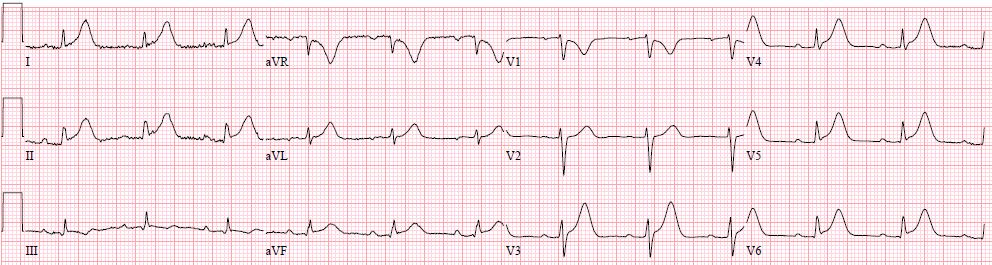
Case Presentation: An 89-year-old female patient with a medical history of atrial fibrillation, TIA, and colon cancer stage IV for which she started Capecitabine three days before her presentation. She presented to the emergency department with intermittent chest pain that started the next day of starting Capecitabine. She was afebrile; her respiratory rate was 18, and her oxygen saturation was 96% on RA. Heart rate was 70, and BP was 111/80. Clinical examination revealed JVP 7 cm and an accentuated component S1. Blood work was significant for neutropenia 4100, thrombocytopenia 149 K, and negative troponin. Chest X-ray showed no significant abnormalities. An initial EKG was done while the patient had no chest pain, revealing normal sinus rhythm without ischemic changes Fig 1. The patient had an episode of chest pain before being discharged from the ED. A repeat EKG was done that showed an acute inferior STEMI Fig 2 and troponin was still negative. She received ASA, Ticagrelor, and sublingual Nitroglycerine, after which her chest pain resolved. The patient had an emergent left cardiac catheterization that showed mild CAD with a patent RCA. An echocardiogram showed a left ventricular ejection fraction of 60% without any wall motion abnormalities. The presumed explanation of the patient’s presentation was coronary vasospasm induced by initiating Capecitabine. The patient’s symptoms and EKG changes resolved after holding Capecitabine.
Discussion: In hospital medicine practice, we deal daily with various case presentations and new emerging medications with a wide range of efficacy, side effects, and interactions. Capecitabine, an oral prodrug of 5-fluorouracil (5-FU), was approved for advanced breast cancer in 1998, then for early-stage and advanced colorectal cancer in April 2001(1). The cardiotoxicity of 5-FU is well-established in the literature (2). However, cardiac adverse effects of capecitabine on coronaries are poorly described. Our case represents a case of coronary vasospasm induced by Capecitabine, with symptoms starting three days within initiating the treatment. EKG showed the picture of Inferior STEMI, and cardiac catheterization showed no significant coronary disease. Based on our PubMed literature review, less than ten cases of Capecitabine-induced coronary vasospasm were reported since 1998. The patients usually present with unstable angina symptoms within 3 days of starting the treatment (3). The patients may have EKG ST segment changes suggestive of acute myocardial infarction or ischemia with negative troponin and clean coronaries (4). These changes usually resolve when the patient is asymptomatic and with nitroglycerine. Capecitabine cardiotoxicity is thought to occur from the action of 5-FU on the endothelium producing endothelin-1 and subsequent coronary vasospasm (5).
Conclusions: Capecitabine-induced coronary vasospasm can happen regardless of pre-existing CAD within a few days of initiating the treatment. Coronary vasospasm is reversible on discontinuation of Capecitabine. Early recognition of this side effect by the hospitalist and discontinuation of Capecitabine in similar cases will help prevent adverse events and cardiotoxicity.