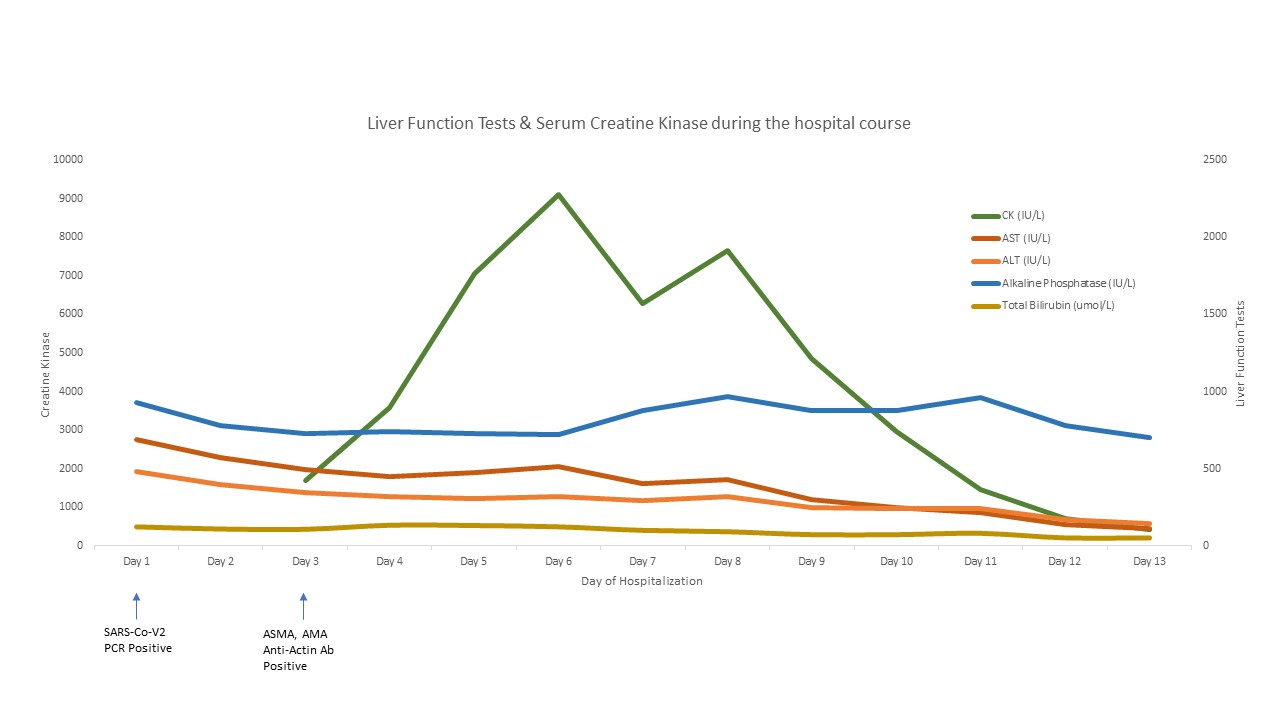
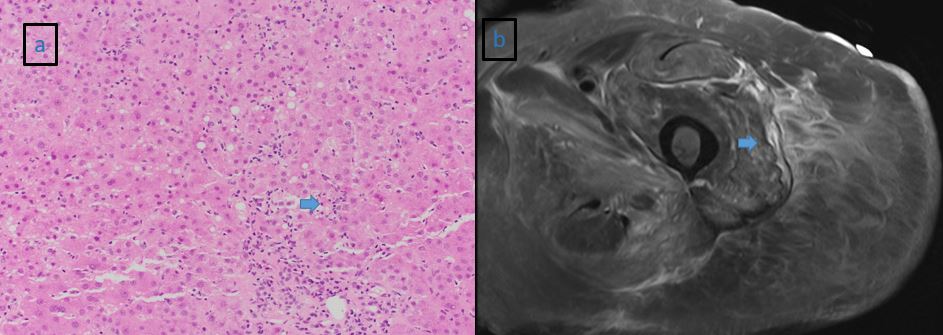
Case Presentation: An 87-year-old female with a history significant for hyperlipidemia on high-intensity atorvastatin for 6 months, multinodular goiter, Raynaud’s disease, spinal stenosis with peripheral neuropathy presents with 3 days of decreased appetite and bilateral lower extremity pain. Her COVID RT-PCR screen was positive, but the patient denied cough, shortness of breath, fever, or diarrhea. Physical exam was significant for painless jaundice and pelvic girdle muscular weakness. Initial labs showed elevated AST 687 IU/L, ALT 479 IU/L, Alkaline phosphatase 931 IU/L, Total bilirubin 7.2 mg/dl, creatinine 1.58 mg/dl, and CK 1688 IU/L (Fig. 1). CT abdomen was negative and MRCP was notable for nonspecific periportal edema. Autoimmune workup was positive for Anti-smooth muscle Ab, Anti-actin Ab, and anti-mitochondrial Ab. Myositis Ab panel and malignancy workup was negative. MRI femur showed autoimmune disease or statin related myositis (Fig. 2). She underwent muscle and liver biopsies for worsening hepatitis and rhabdomyolysis. Her liver biopsy demonstrated an acute hepatitis pattern of injury with a differential diagnosis of viral hepatitis or drug induced liver injury (Fig. 2). Her muscle biopsy showed myophagocytosis due to rhabdomyolysis from a viral disease vs. drug induced adverse event. Our patient was managed for acute liver and muscle injury from statin toxicity exacerbated in the setting of the COVID-19 viral syndrome with discontinuation of the statin therapy, and with cautious fluid boluses. Her CK peaked at 9092 IU/L and then started improving along with her LFTs. (Fig. 1) On post-discharge clinic follow up her labs had normalized.
Discussion: It is theorized that SARS-CoV-2 directly binds to ACE2 positive cholangiocytes and creates a cytotoxic effect and/or causes a “cytokine storm” dysregulation of the innate immune response 1. Major histological changes in patients with altered LFTs include steatosis, acute hepatitis, and portal inflammation 2. Statin induced hepatotoxicity commonly occurs in the first 3 months of therapy, is dose-dependent, includes hepatocellular and cholestatic patterns of injury, and rarely can trigger autoimmune hepatitis in susceptible patients which can progress to chronic liver failure. Most patients recover with the cessation of offending statin medication but close monitoring to initiate immunosuppressive therapy is warranted until complete recovery occurs 3. The exact pathophysiology of rhabdomyolysis in COVID-19 has not yet been studied, but viral induced rhabdomyolysis is hypothesized to be related to direct viral invasion of muscle tissue or cytokine mediated destruction and, can also be exacerbated by other known causes of rhabdomyolysis, such as drug interactions 4,5. While the treatment of rhabdomyolysis is fluid resuscitation and avoidance of any potential offending agents, small boluses of fluids are suggested, especially when treating patients with known COVID-19 infections that are at risk of acute respiratory failure 5.
Conclusions: Discontinuation of the inciting agent as well as recovery from the viral illness resulted in the resolution of the abnormalities in our patient. The evidence is inconclusive as to whether the patient’s acute liver injury and rhabdomyolysis were a result of her statin usage, COVID-19 infection, or a worsened picture given the combination of both. This diagnostically nuanced case highlights some of the challenges faced during a pandemic where the clinical disease spectrum of SARS-CoV-2 continues to evolve.