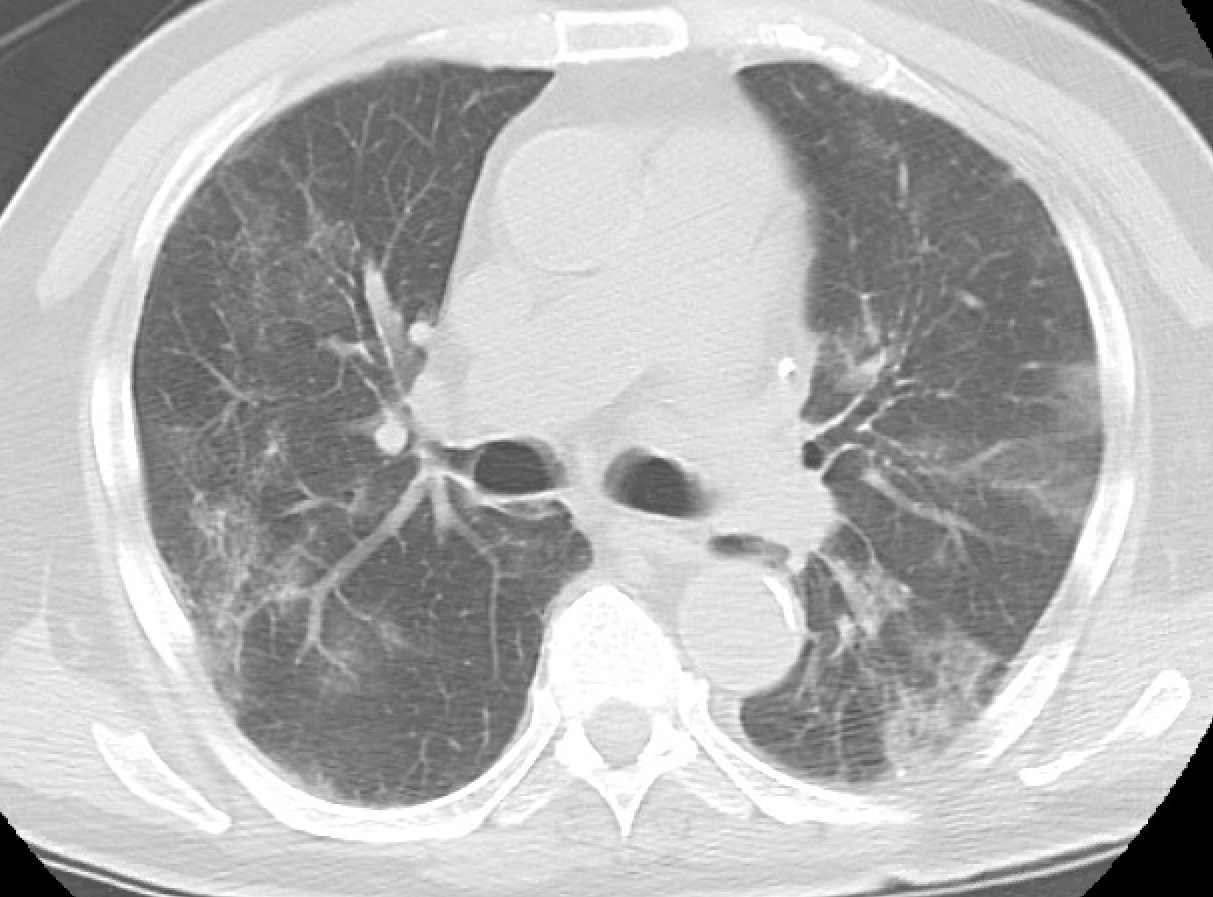
Case Presentation: This is an 84-year old Caucasian man with a past medical history of hypercholesterinemia who developed dry cough, mild shortness of breath, generalized weakness, and fever 13 days prior to the presentation. Three days after onset, he was tested positive for SARS-CoV-2 virus. His shortness of breath continued to worsen, and the patient was hypoxic upon arrival to the hospital, requiring 4 liters/min of supplemental oxygen. Physical exam revealed scleral icterus. Laboratory work was significant for severe anemia (hemoglobin – 4.4 g/dL) and indirect bilirubinemia (2.3 mg/dL). CT scan of chest, abdomen and pelvis did not show any occult hemorrhage but revealed diffuse patchy bilateral ground-glass opacities within the lungs and splenomegaly. The patient was found to have positive direct Coombs test with anti-K antibodies and IgG pan-agglutinins. He received packed RBCs that were type-specific, K-negative, and “least incompatible” based on cross-match. Further analysis showed lactate dehydrogenase of 1253 U/L, haptoglobin <10 mg/dL, and reticulocyte count of 120x109/L. Peripheral smear identified numerous nucleated RBCs and microspherocytes. Given the presence of warm antibody-mediated hemolytic anemia, he was diagnosed with warm AIHA secondary to Coronavirus disease 2019 (COVID-19). The patient was started on convalescent plasma therapy, remdesivir, and dexamethasone. In the first 24 hours, he received 5 units of packed RBCs and remained stable, with hypoxia and dyspnea improving significantly on the second day.
Discussion: Recognizing AIHA in the setting of COVID-19 is important to avoid delay in treatment. Warm autoimmune hemolytic anemia (AIHA) is a rare autoimmune disorder mediated by autoantibodies that are active at normal body temperature. It is commonly associated with underlying conditions, such as viral infections, autoimmune disorders, lymphoproliferative diseases, immunodeficiency states, and pregnancy. There are only a few case reports of AIHA in COVID-19 patients. Physical exam may show jaundice and splenomegaly. In addition to low hemoglobin, laboratory evaluation may be notable for increased reticulocyte count, elevated lactate dehydrogenase, low haptoglobin, indirect bilirubinemia, and spherocytosis/microspherocytosis. Diagnosis is made based on presence of hemolytic anemia mediated by warm antibodies. Due to the presence of pan-reacting autoantibodies, identifying cross-matched blood products may not be possible. If severe anemia is present, clinicians should contact blood bank immediately for type-specific, “least incompatible” blood products. Glucocorticoids have been shown to be effective for management of AIHA in addition to transfusion. Since glucocorticoids are also used in moderate to severe COVID-19 cases, it is reasonable to treat COVID-19 associated AIHA with glucocorticoids.
Conclusions: Warm AIHA is a rare complication of COVID-19 that should be recognized as soon as possible. It is crucial to notify blood bank for urgent transfusion with “least incompatible” blood products because the autoantibodies can delay the crossmatch. Glucocorticoids treatment provides benefit to both AIHA and COVID-19.