Background: Overnight vital sign assessment can disrupt sleep in the hospital and may be unnecessary in clinically stable patients. However, providers may not feel comfortable determining which patients can safely forego overnight vitals. We studied the effect of a clinical decision support (CDS) tool embedded in the electronic health record (EHR) that automatically identified clinically stable patients and encouraged providers to defer overnight vital sign checks for those patients.
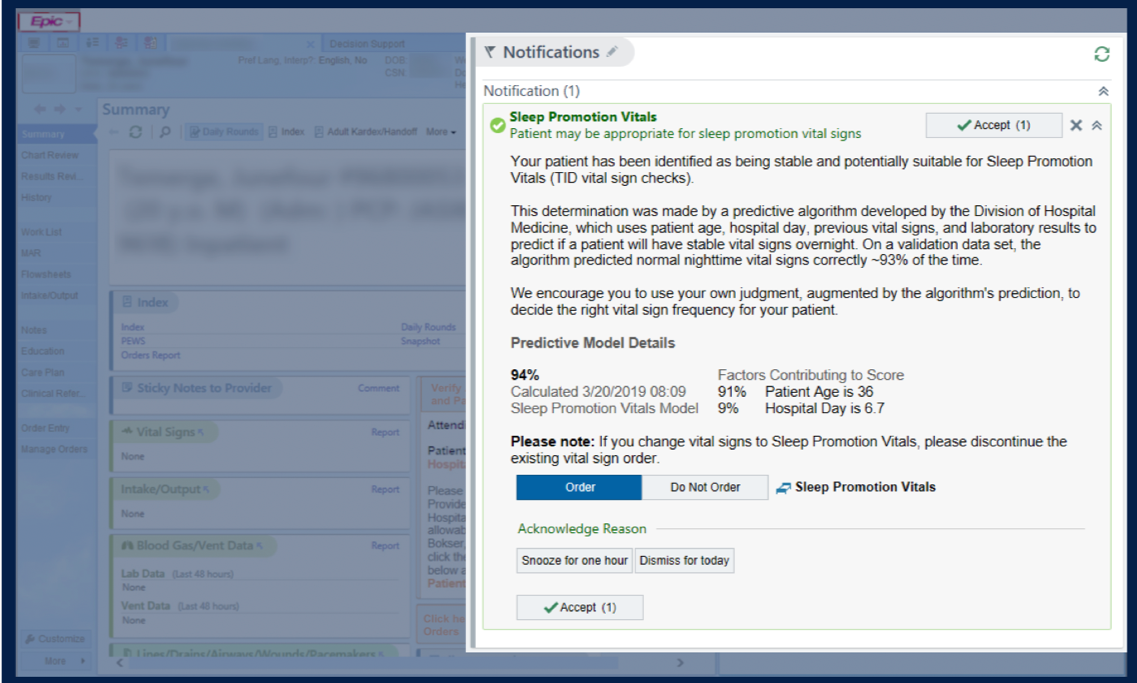
Methods: This was a pragmatic randomized controlled trial (RCT) conducted at a tertiary academic hospital from March 2019 to September 2019. We first developed an algorithm that could identify patients with a high probability of stable overnight vital signs. This algorithm was then incorporated into the Epic-based EHR and applied to all adults admitted to the general internal medicine service. Patients identified by the algorithm as “stable” were then randomized to either the intervention or control arm. The intervention comprised of displaying our CDS notification in the EHR when the primary provider for that patient opened the patient’s electronic chart. The charts for patients in the control arm did not show the notification (providers could still manually opt to forego overnight vital sign checks). The CDS tool was a novel side-bar notification that 1) alerted the provider that the patient was identified by our algorithm as having a greater than 90% chance of having stable nighttime vital signs, 2) displayed the data that led to this prediction, 3) suggested reducing vital sign checks if the provider agreed, and 4) allowed the provider to reduce vital sign checks from within the alert by ordering “sleep-promotion vitals” (SPV), which were TID vitals (Figure 1).The primary outcome was average nightly sleep opportunity, an EHR-derived metric that measures the longest period of time between recorded nighttime interruptions (e.g. blood pressure check, scheduled medication administration, etc.). Secondary outcomes were delirium, as measured by the Nursing Delirium Screening Scale (NuDESC), and process measures, such as the number of SPV orders placed and the number of overnight vital sign checks done. To evaluate for potential harms, we monitored balancing outcomes of rapid response calls, ICU transfers, code blues, and deaths.
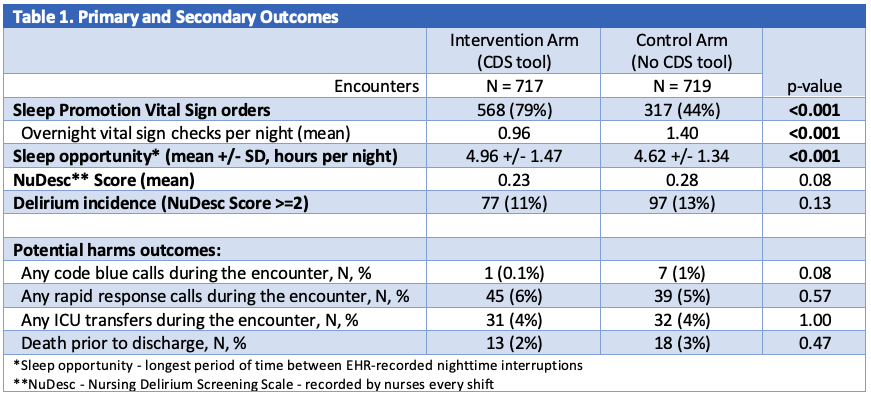
Results: A total of 1436 patient encounters were randomized over the trial period, 717 to the intervention arm and 719 to the control arm. Sleep-promotion vitals were ordered in 568 encounters (79%) in the intervention arm, compared to 317 (44%) in the control arm (p < 0.001). Patients in the intervention arm had an average of ~20 minutes additional sleep opportunity per night compared to the control arm (4.96 vs. 4.62 hours, p < 0.001). In 35% of encounter nights, overnight vital signs were checked despite an SPV order in place. Delirium incidence, as defined by a NuDESC score greater than or equal to 2, was not statistically different between the two arms, though there was a tendency towards lower mean NuDESC scores in the intervention arm (0.23 vs 0.28, p=0.08). There were no significant differences in harm events (Table 1).
Conclusions: We developed a CDS tool that used predictive analytics to help providers identify stable hospitalized patients that could forego overnight vital sign checks. Our tool was effective in changing clinician behavior (nearly doubling SPV orders), and, despite a high rate of order noncompliance, increased average sleep opportunity by 20 minutes without obvious harm.