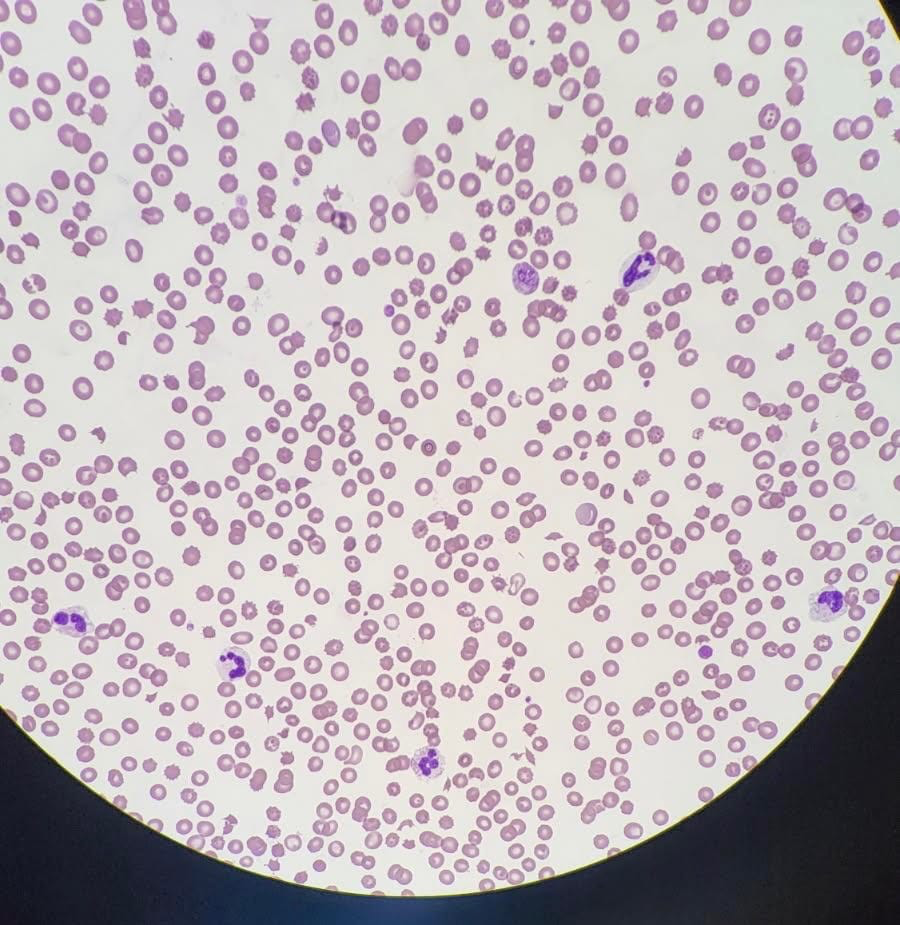
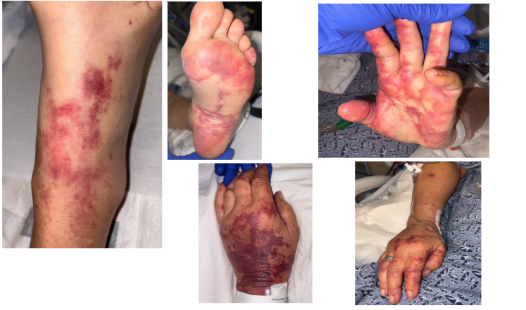
Case Presentation: This is a case of a 73-year-old woman with a past medical history of hypothyroidism, hyperlipidemia, depression/anxiety, and remote splenectomy who developed acute nausea and vomiting while traveling cross‑country on a road trip. She presented in shock with hypotension, tachypnea, and hypoxia. Initial studies revealed acute kidney injury, high anion gap metabolic acidosis, thrombocytopenia, and anemia. Hemolysis workup revealed an elevated LDH at 3,042, haptoglobin < 30, and evidence of schistocytes on peripheral smear, consistent with microangiopathic hemolytic anemia. Blood cultures grew alpha‑hemolytic streptococci. Despite broad-spectrum antibiotics, her renal function deteriorated, requiring renal replacement therapy, and thrombocytopenia worsened. Given progressive end‑organ injury, plasma exchange was initiated for suspected thrombotic microangiopathy (TMA). ADAMTS13 activity returned at 20%, not indicative of severe deficiency typically seen in TTP, and complement C3 was low. She was transferred for hematology evaluation, diagnosed with atypical hemolytic uremic syndrome (aHUS), and ultimately treated with eculizumab.
Discussion: This case illustrates the diagnostic complexity of thrombotic microangiopathies and highlights the importance of rapidly distinguishing aHUS from TTP, as management strategies diverge significantly. While both present with MAHA and thrombocytopenia, this patient’s predominant renal failure, absence of neurological deficits, incomplete reduction in ADAMTS13 activity, and depressed complement levels pointed toward complement‑mediated aHUS rather than primary ADAMTS13 deficiency. Infection-associated complement activation likely served as the trigger. Early initiation of plasma exchange provided critical stabilization while laboratory data were pending, reinforcing the need for prompt empiric management when severe TMAs are suspected. Recognition of complement pathway dysregulation is essential, as timely initiation of terminal complement blockade with agents such as eculizumab can be organ‑saving.
Conclusions: Atypical HUS should remain a key consideration in adults presenting with severe TMA, particularly when ADAMTS13 activity is reduced but not severely deficient and renal dysfunction is prominent. This case underscores the substantial clinical overlap between TTP and aHUS and highlights practical diagnostic clues that can guide early decision‑making. For hospitalists—often the first to evaluate these critically ill patients—maintaining a high index of suspicion, initiating supportive care including dialysis and plasma exchange when appropriate, and promptly pursuing targeted diagnostic testing are essential to improving outcomes in complement‑mediated TMA.