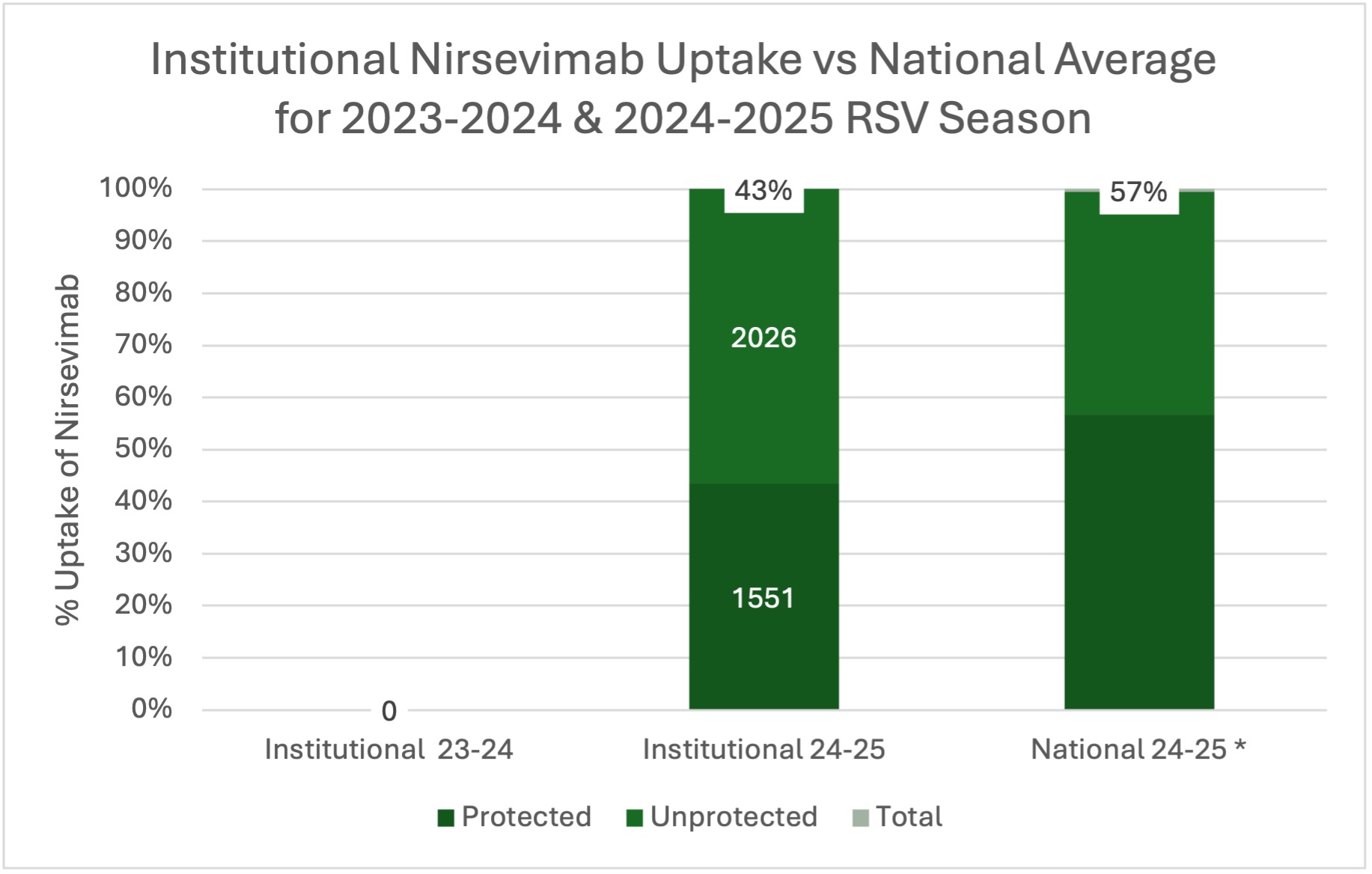
Background: Respiratory syncytial virus (RSV) is a leading cause of acute lower respiratory infection in infants, responsible for approximately 58,000–80,000 hospitalizations and 100–300 deaths annually among U.S. children under 5 years, with the highest hospitalization rates in infants under 6 months 1. In 2023, the FDA approved nirsevimab, a long-acting monoclonal antibody administered as a single intramuscular dose, for all infants under eight months entering or born during epidemiologic RSV season (fall through winter), as well as for children 8-19 months entering their second RSV season if they meet high-risk criteria 2. Following the introduction of nirsevimab and maternal vaccination, RSV-associated hospitalization rates in eligible infants decreased by 43% (RSV-NET data) and 28% (NVSN data) during the 2024–2025 RSV season compared to pre-intervention seasons (2018–2020), with the largest reduction in infants aged 0–2 months 3. Despite its efficacy, early data shows variability in uptake across health systems. We implemented a structured administration protocol with pediatric provider education to increase eligible infants receiving nirsevimab during our first available RSV season 2024-2025. National uptake for immediately following the 2023–2024 RSV season was estimated at 44.6% 3. We aimed to increase uptake at our institution from 0% in 2023–2024 to 45% in 2024–2025.
Methods: A retrospective, IRB-approved review included pediatric patients aged 0-24 months seen at our pediatric outpatient sites between October 1, 2024 and May 31, 2025. Manual chart review through the EMR identified demographics, high-risk conditions, and nirsevimab administration. Exclusion criteria included age greater than 24 months, prior RSV prophylaxis, or maternal RSV vaccination within 2 weeks prior to delivery. De-identified data were analyzed to determine uptake rates, and analyzed by chi-squared test.
Results: Among 3576 eligible patients, 1551 (43.3%) received nirsevimab. Preliminary review of 2026 infants who did not receive nirsevimab revealed that among a single site subset of 585, 77 had vaccinated mothers. Compared to eligible infants in the prior season, there was a change of 43.3% (χ2= 1584, p < 0.0001) during the 2024-2025 RSV season.
Conclusions: Institutional uptake of nirsevimab for the 2024-2025 RSV season aligned with national trends from the previous season. Updated data for the national 2024-2025 RSV season estimates that both maternal vaccination and nirsevimab combined protected 57% of eligible infants 4. Although local maternal vaccination rates were not captured at our institution, nirsevimab uptake increased significantly towards our goal. Accounting for known maternal vaccination, total overall RSV protection at our institution further increases. These findings show how targeted and structured education can result in increased recommendation and uptake of nirsevimab, contributing to larger public health efforts. Data quality issues, including inconsistent EMR documentation and order-diagnosis associations, were identified as key barriers to accurate surveillance and improvement tracking. Future efforts will focus on improved documentation, continued provider and patient education to further optimize RSV protection, as well as cross-site communication to improve real-time uptake tracking for both infants and mothers.