Background: Almost three-quarters of hospitals with 50 or more beds have a palliative care program, and the percentage of annual hospital admissions receiving an initial palliative care consultation is more than 5%. Methylnaltrexone (MNTX) is a peripherally acting µ-opioid receptor antagonist indicated for opioid-induced constipation (OIC) in patients with advanced illness who are receiving palliative care and in patients with chronic noncancer pain. We investigated if baseline patient characteristics impact the safety and efficacy of MNTX in those with advanced illness and OIC.
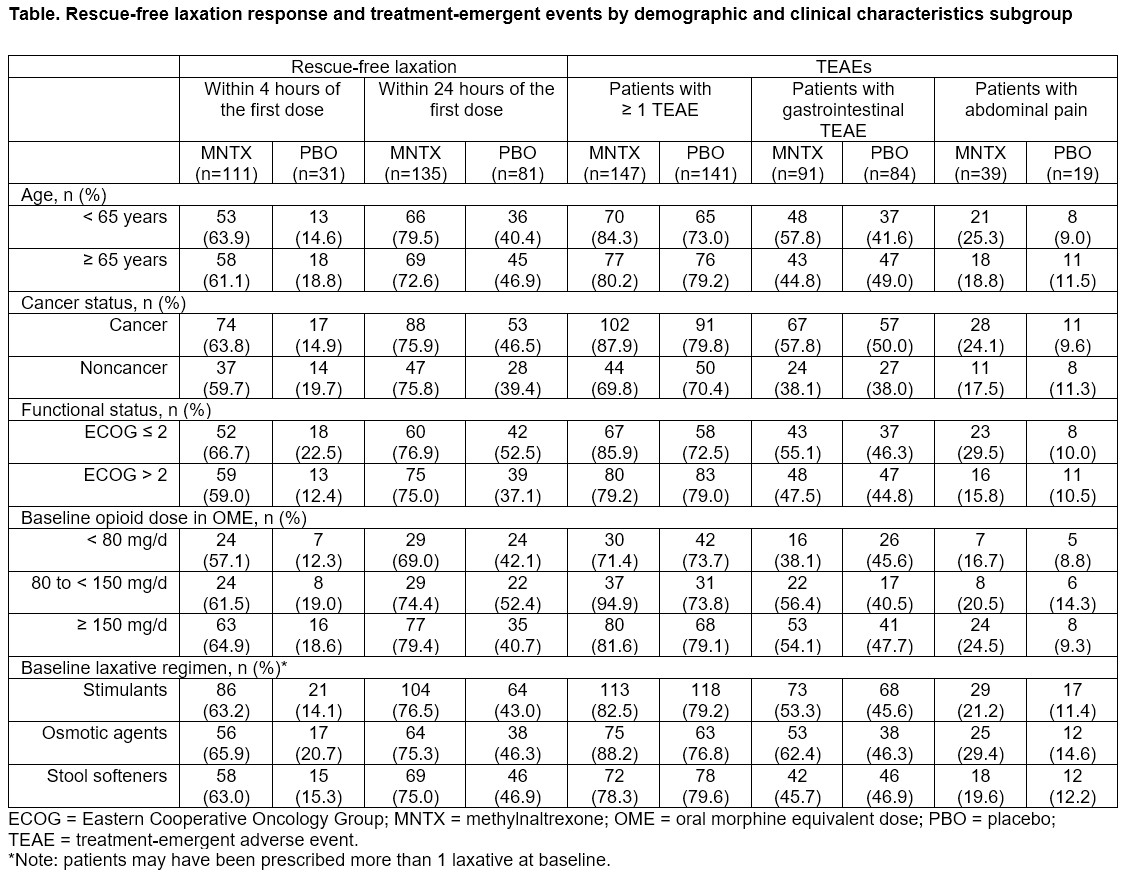
Methods: This analysis pooled data from 2 randomized, double-blind, placebo (PBO)-controlled studies (study 302–NCT00402038; study 4000‒NCT00672477) in patients with advanced illness, including cancer. Study 302: Patients were randomized to receive subcutaneous (SC) MNTX 0.15 mg/kg or PBO every other day (QOD) for 2 weeks, with possible dose escalation to 0.30 mg/kg at week 2. Study 4000: Patients received weight-based SC MNTX 8 mg or 12 mg QOD or PBO for 2 weeks. The proportion of patients achieving a rescue-free laxation within 4 or 24 hours after the first dose of study drug was assessed in patient subgroups stratified by baseline age (< 65 vs ≥ 65), Eastern Cooperative Oncology Group (ECOG) status (≤ 2 vs > 2), cancer status, laxative type (osmotic agents, stimulants, stool softeners), and opioid requirement (oral morphine equivalent dose < 80 mg/d, 80 ‒ < 150 mg/d, and ≥ 150 mg/d). Treatment-emergent AEs (TEAEs), gastrointestinal (GI) TEAEs, and abdominal pain were evaluated.
Results: Overall, 363 patients were included in this analysis (MNTX = 178; PBO = 185). Mean age was 67.8 years in the MNTX group and 66.7 years in the PBO group; 46.8% were men across all groups. A significantly greater proportion of patients receiving MNTX achieved rescue-free laxation within 4 hours (n = 111/178, 62.4% vs n = 31/185, 16.8%, P < .0001) and 24 hours (n = 135/178, 75.8% vs n = 81/185, 43.8%, P < .0001) of the first dose vs PBO. These trends were consistent across all subgroups (Table). Most patients experienced ≥ 1 TEAE in the overall population (MNTX 82.1%; PBO 76.2%); results were similar when stratified by baseline cancer, ECOG, opioid, or laxative group (Table). More than half of TEAEs were GI in nature; abdominal pain was more common in patients receiving MNTX across subgroups.
Conclusions: MNTX treatment was superior to PBO in achieving RFL within 4 and 24 hours after the first dose, irrespective of patients’ cancer status, baseline ECOG status, or baseline opioid or laxative use. MNTX remained consistently safe across different baseline clinical and demographic characteristics.