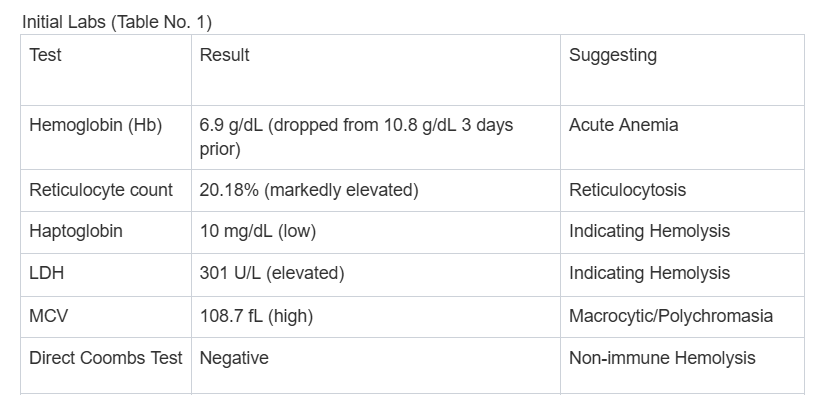
Case Presentation: A 44-year-old male with poorly controlled diabetes and orthostatic hypotension presented with worsening dizziness. Three days pre-admission, he started metformin, pioglitazone, and glipizide for diabetes management. In the ED, the patient was tachycardic and hypotensive, responding well to IV fluids. Initial labs (as shown in Table No. 1) suggested acute, non-immune hemolytic anemia.{Table No. 1 Here}Hemoglobin electrophoresis, LFTs, Iron panel, B12, folate, Coagulation panel, homocysteine & MMA, and TSH were unremarkable. HIV, Direct Coombs Test, PNH flow cytometry, cold agglutinins, and cryoglobulin were negative. The patient received 1 unit of packed red blood cells and all oral antihyperglycemic agents were stopped. His hemoglobin stabilized (8.8–9.4 g/dL). Subsequent testing showed low G6PD enzyme activity (33 U/dL). The patient then recalled a prior diagnosis of G6PD deficiency during army screening.
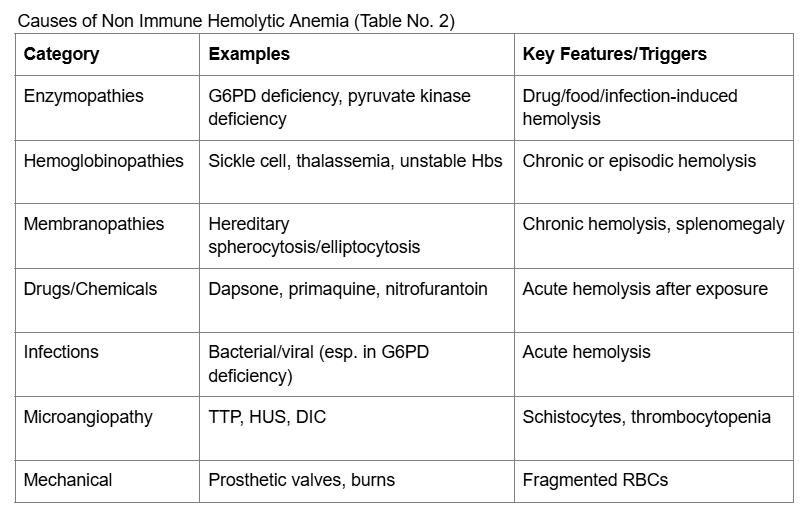
Discussion: This case highlights challenges related to readmission, transition of care, and patient safety. Acute hemolytic anemia in G6PD deficiency typically develops within 24–72 hours of exposure to triggers. 1 The temporal association between the initiation of glipizide and the onset of acute hemolysis suggests the sulfonylurea was the likely cause. Although large cohort studies indicate the overall risk of major hemolysis requiring hospitalization is very low (0.2%), primarily triggered by fava beans or infection, case reports confirm that sulfonylureas, including glipizide, can precipitate acute hemolysis in susceptible individuals. 2 Metformin and pioglitazone were deemed unlikely culprits due to their safety profiles and lack of mechanistic plausibility as oxidative stressors. Other causes of non-immune hemolysis (as shown in Table No. 2) were excluded by laboratory testing and clinical presentation.{Table No. 2 Here}Clinicians should be aware that acute hemolysis can falsely lower HbA1c, complicating diabetes management and potentially suggesting G6PD deficiency in patients with a disproportionately high fasting blood glucose to HbA1c ratio.3 It is difficult to say if this reaction was idiosyncratic or an oxidative stress in the setting of G6PD deficiency. All hemolysis reported with sulfonylureas have consistently involved patients with undiagnosed G6PD deficiency. Given the unpredictable nature and potential severity of this reaction, re-challenge with sulfonylureas after a hemolytic episode is not considered safe. Guidelines and expert consensus recommend permanent discontinuation of the offending agent and selection of alternative antihyperglycemic therapies in affected patients.
Conclusions: Sulfonylurea-induced hemolysis in G6PD deficiency is a rare but significant, acute adverse drug reaction. Notably, all reported hemolytic reactions occurred in patients with previously undiagnosed G6PD deficiency. Prompt recognition and permanent withdrawal of the suspected oxidizing agent are crucial for optimal outcomes, as re-challenge is unsafe. This case critically reminds hospitalists to inquire about G6PD deficiency before initiating medications that may trigger oxidative stress, even those (like glipizide) with extremely low overall risk.