Background: Patients admitted to New Hanover Regional Medical Center with a history of opiate use disorder (OUD) and serious infections require a median length of stay (LOS) of 33 days and discharge against medical advice (AMA) 26% of the time, resulting in quality and safety gaps which includes incomplete treatment, readmissions, and high cost of care. While standard of care for systemic staphylococcal infections consists of partial outpatient antibiotic regimens, OUD patients are not offered outpatient treatment due to perceived risk of injection attempts through indwelling catheters. Our hypothesis was that patients with OUD and serious infections who have an alternative option of weekly outpatient antibiotic infusions would show an improvement in treatment completion rates.
Methods: A multidisciplinary team consisting of hospitalists, infectious disease consultants, and members of the “Code Outreach” substance disorder team used LEAN principles to determine eligibility criteria for use of weekly dalbavancin infusions in our vulnerable population. Inclusion criteria consisted of: (1) staphylococcal infection sensitive to vancomycin, (2) osteomyelitis or skin/soft tissue infection, (3) completed course of therapy for bacteremia, (4) easy vascular access, (5) limited or no comorbid conditions, (6) engaged in treatment for OUD, and (7) stable housing. Patients were excluded if they had endocarditis or an undrained abscess. The pilot program was presented to the Antibiotic Stewardship Committee and the Pharmacy and Therapeutics team for review given the higher cost of dalbavancin compared to alternatives.
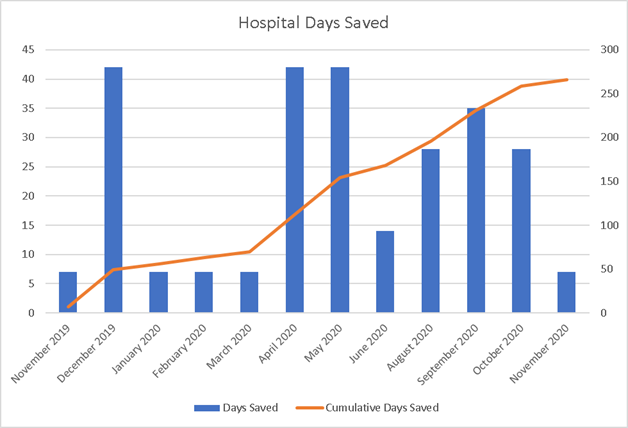
Results: From November 2019 to November 2020, 19 patients who met eligibility criteria were discharged with the plan to complete outpatient therapy with dalbavancin. The average age enrolled was 37 years; 47% were female. MSSA was isolated in 7 patients (37%), MRSA in 9 patients (47%), and the organism was presumed staphylococcal in 3 patients (16%). Three patients (15.8%) left AMA; one patient still completed outpatient therapy, one received 3 of 5 outpatient doses, and one was lost to follow up. Treatment plans included outpatient administration of dalbavancin at the infusion center for 13 patients (68%); other patients received a single dose of dalbavancin prior to discharge. No pilot patients readmitted to our facility since discharge; one patient who left AMA was lost to follow up and expired. The average LOS for included patients was 13 days. Successful outpatient treatment saved 266 cumulative bed-days which is an estimated savings of $513,646 based on the 2016 American Hospital Association Annual Survey for North Carolina non-profit expenses per inpatient day. Cost of administration of dalbavancin for these 19 patients was estimated at $25350, for a total estimated savings of $488,296.
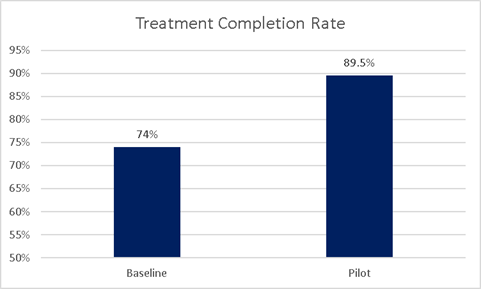
Conclusions: Implementation of weekly dosing of dalbavancin in patients with OUD and serious infections resulted in a significant improvement in treatment completion rates from 74% to 89.5%. Hospital LOS in our pilot population decreased by 60%, saving 266 bed-days and over $488,000; considering that over one quarter of patients in the baseline population left AMA, this calculation underrepresents the theoretical days saved if those patients had completed therapy. This pilot substantiates the hypothesis that partial outpatient antibiotic treatment regimens are a safe and cost-effective mechanism for completing treatment for serious infections in patients with OUD.