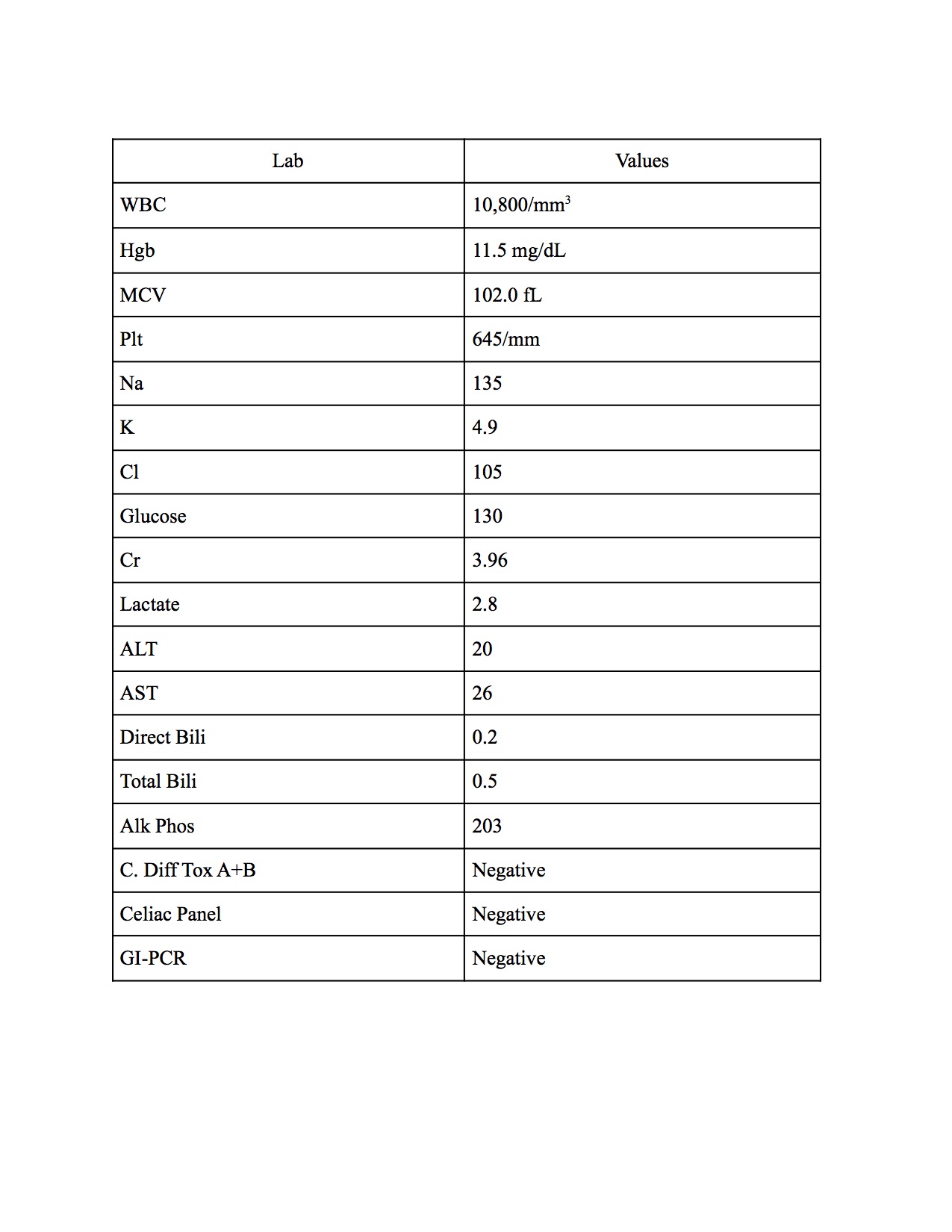
Case Presentation: A 78-year-old female with a medical history of deep vein thrombosis, ovarian cancer status post resection and currently in the TESARO trial (receiving Dostarlimab and Niraparib or placebo; last dose was 3 months ago) presented to the emergency department from her outpatient provider due to inability to obtain a blood pressure reading in the setting of increased lethargy, fatigue, and chronic diarrhea. She reported non-bloody, watery, loose stools for the last 3 months (about 3-5 episodes a day) since she completed the last dose of the trial medication she has been on for her ovarian cancer. Patient denied any sick contacts, recent travel, or different food intake. She also stated she has not had a colonoscopy for more than 5 years. Vitals signs were notable for a blood pressure of 69/38 mmHg. Laboratory evaluation revealed thrombocytosis and macrocytosis (Table 1). CT of the abdomen/pelvis was suggestive of possible ileus; however, a degree of small bowel obstruction was not excluded. Patient was started on Loperamide and her diarrhea greatly improved. Colonoscopy revealed increased mucosal vascular pattern in the entire colon and diverticulosis in the sigmoid colon. Multiple biopsies obtained throughout the colon revealed chronic colitis with marked thickening and irregularity of the sub epithelial collagen plate, consistent with collagenous colitis. Patient was started on budesonide 9 mg for 9 weeks for treatment of PDL1 inhibitor mediated microscopic colitis.
Discussion: Immune-mediated colitis (IMC) is an immune-related adverse event in patients treated with immune checkpoint inhibitors (ICI/ICPIs). Management of IMC greatly depends on classification of symptoms and is further divided into grades 1 through 4. Grade 1 is defined as less than 4 bowel movements per day above baseline and patients undergo symptomatic treatment of diarrhea while continuing ICI treatment. For patients with grade 2 colitis defined as 4-6 bowel movements per day along with at least one other symptom such as abdominal pain or hematochezia, ICIs are discontinued and treatment with prednisolone or oral budesonide is initiated. In grade 3 or 4 toxicity defined as more than 7 stools above baseline per day or with signs consistent with possible bowel perforation, ICIs should be discontinued and patients should most likely be hospitalized to receive intravenous fluids and systemic corticosteroid treatment. Patients who had grade 4 toxicity should permanently discontinue ICIs. All other patients could potentially resume ICI treatment if they continue to remain symptom free. While there are many similarities between IMC and inflammatory bowel disease (IBD), IMC is usually indicative of acute colitis rather than chronic colitis. In our case, this patient was categorized into grade 2 given the increased occurrence of diarrhea with no other life-threatening signs. Patient was started on a taper of oral budesonide 9 mg as treatment guidelines suggest. ICI treatment was resumed after steroids were tapered given that patient remained symptom free.
Conclusions: Patients suffering from immune mediated colitis are often treated with a corticosteroid regimen based on the severity of colitis. IMC usually differs from IBD in that it is usually a form of acute colitis rather than chronic colitis, however this may not always be the case. Heightened awareness and consideration for colitis secondary to ICI treatment may promote timely detection, symptomatic treatment, and resolution of the colitis before it has the chance to become severe.