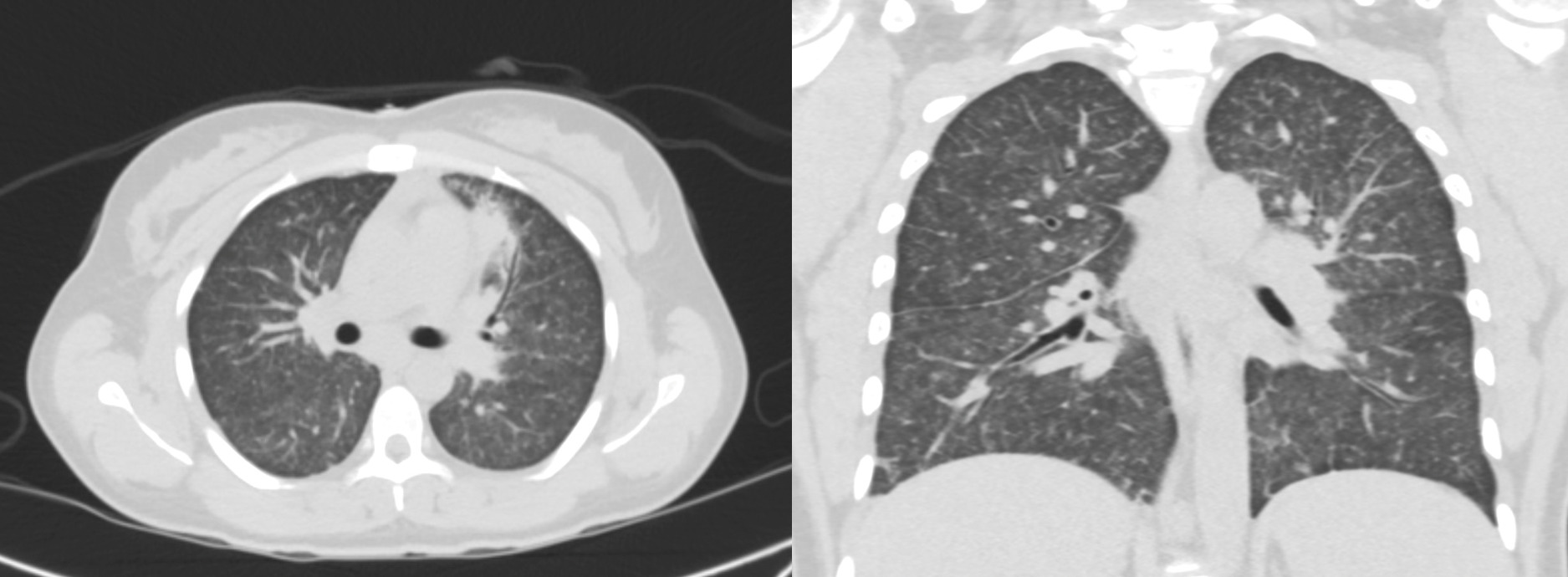
Case Presentation: A 33-year-old post–kidney transplant woman with Type 1 Diabetes presented with several days of persistent fever, chills, mild headache, photophobia, dry cough, fatigue, and night sweats. She had recently moved from Tennessee to Florida a couple of months prior. On admission, her vital signs and physical examination were unremarkable, and no meningeal signs were present. CT head and MRI brain were both negative. Because of concern for meningitis, she was initially started on empiric antimicrobials including vancomycin, cefepime, acyclovir, and ampicillin. However, a negative lumbar puncture ruled out a neurological infection. A CT scan of the chest showed diffuse miliary nodules throughout the lungs and focal consolidation/pneumonia in the lingula. Despite an indeterminate beta-D-glucan, urine Histoplasma antigen was positive. The diagnosis of histoplasmosis was further supported by positive next-generation sequencing. She was started on liposomal amphotericin B as well as empiric antitubercular therapy. Antitubercular therapy was discontinued after a negative Quantiferon test. After one week of amphotericin B, itraconazole was initiated. Her condition improved, and she was discharged on itraconazole therapy to be continued for 12 weeks. Tacrolimus was continued, while mycophenolate mofetil was held.
Discussion: Clinical presentation of histoplasmosis is often nonspecific and frequently asymptomatic, but it may manifest with persistent fever, weight loss, and pancytopenia. Pulmonary involvement is common and typically appears as diffuse reticulonodular or miliary infiltrates on high-resolution computed tomography. Extrapulmonary disease may affect the liver, spleen, bone marrow, central nervous system (CNS), and gastrointestinal tract. Neurological manifestations can include headache, fever, nausea, vomiting, and focal neurological deficits.Diagnosis is primarily made through Histoplasma antigen testing, which can detect antigen in urine, serum, bronchoalveolar lavage (BAL), and cerebrospinal fluid (CSF), and is considered the most sensitive method. Fungal cultures obtained from blood, bone marrow, or respiratory samples remain the gold standard for definitive diagnosis. Molecular techniques, including polymerase chain reaction (PCR) and DNA probe assays, can also detect Histoplasma DNA in tissue and body fluids.Treatment generally consists of liposomal amphotericin B at 3–5 mg/kg intravenously for one to two weeks, followed by oral itraconazole at 200 mg three times daily for three days and then 200 mg twice daily to complete a total of 12 weeks of therapy.Immunocompromised patients are at higher risk of complications after acute pulmonary histoplasmosis, including chronic pulmonary histoplasmosis, progressive disseminated disease, mediastinal fibrosis, histoplasmoma, and broncholithiasis. Mortality among solid organ transplant recipients with histoplasmosis remains as high as 10%.
Conclusions: Histoplasmosis should be considered in immunocompromised patients presenting with nonspecific symptoms, particularly those who have lived in or recently traveled from endemic areas. Although urine antigen testing is a sensitive diagnostic tool, it should be complemented by advanced methods such as next-generation sequencing. Diffuse miliary nodules strongly correlate with pulmonary involvement. Treatment with liposomal amphotericin B followed by itraconazole is effective, with adjustments to immunosuppressive therapy as needed.