Background: Methylnaltrexone (MNTX) is a peripherally acting µ-opioid receptor antagonist indicated for opioid-induced constipation (OIC), which can be a serious consequence of opioid analgesia. We evaluated whether baseline osmotic agent (eg, milk of magnesia, polyethylene glycol, lactulose, sorbitol) or stimulant (eg, senna, bisacodyl) use affects the efficacy and safety of MNTX among a subgroup of OIC patients with advanced illness.
Methods: Two multicenter, randomized, double-blind, placebo-controlled, institutional review board–approved clinical studies in adult patients with OIC and advanced illness were pooled (study 302, NCT00402038; study 4000, NCT00672477). Study 302 compared subcutaneous MNTX 0.15 mg/kg versus placebo and study 4000 compared body weight‒based subcutaneous MNTX 8 mg (38–<62 kg) or 12 mg (at least 62 kg) versus placebo. The number of patients who received baseline osmotic agents and/or stimulants, which were permitted to continue during the studies, were identified. Efficacy endpoints included rescue-free laxation within 4 or 24 hours after the first dose (ie, a spontaneous bowel movement without requiring rescue laxatives) and pain intensity (evaluated using an 11-point scale). Safety endpoints included treatment-emergent adverse events.
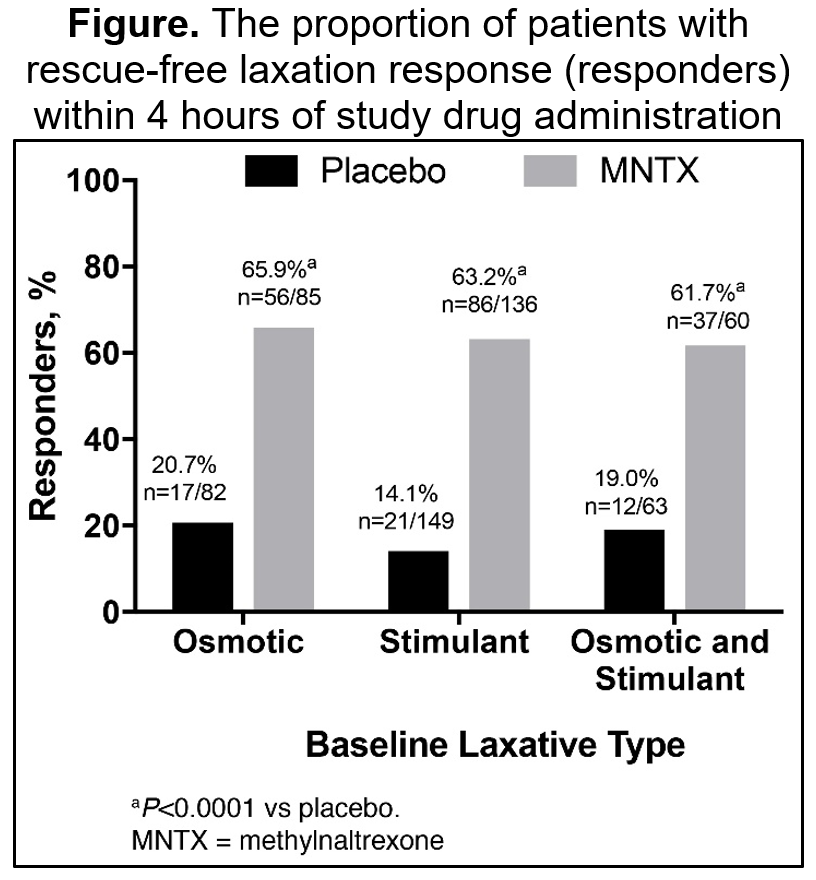
Results: The pooled analysis yielded 330 patients with advanced illness (the most common primary diagnosis being cancer [63.5%] followed by pulmonary [12.2%] and cardiovascular [10.6%] disease) who were using a stimulant and/or an osmotic agent at baseline (stimulant: n=286, osmotic: n=167, stimulant and osmotic: n=123). The pooled baseline demographic population had a mean age of 66.2 years, 50.5% were women, 94.8% were white, and the mean weight was 72.0 kg. A greater proportion of patients who received MNTX (63%–66%) had a rescue-free laxation within 4 hours compared with placebo (14%–21%) regardless of the type of laxative used at baseline (P<0.0001, Figure). Similar findings were observed for those with a rescue-free laxation response within 24 hours of MNTX treatment (72%–77%) versus placebo (43%–46%, P<0.005). There were no significant differences between MNTX or placebo in the change from baseline pain scores after 1 day or 7 days regardless of the type of laxative used at baseline. The most commonly reported treatment-emergent adverse events (>5%) that occurred in more MNTX-treated patients were abdominal pain (both cohorts), flatulence (both cohorts), back pain (both cohorts), asthenia (osmotic cohort only), dizziness (osmotic cohort only), peripheral edema (stimulant cohort only), pyrexia (stimulant cohort only), and falls (stimulant cohort only).
Conclusions: Use of MNTX in patients with advanced illness significantly increased the proportion of patients with a rescue-free laxation response within 4 and 24 hours of treatment in patients refractory to baseline use of an osmotic and/or stimulant laxative. MNTX did not reduce the analgesic effects of opioid treatment and did not induce unexpected adverse events within the cohorts.